Landmark Danish Study: 1.2 Million Children Confirm Aluminum Vaccine Safety |
September 09, 2025 . 4 Minutes read
Conclusive Evidence: Addressing Concerns on Aluminum Vaccine Adjuvant Safety
A groundbreaking Danish study, examining 1.2 million children over 24 years, has delivered very reliable and conclusive evidence on the safety of aluminum adjuvants in childhood vaccines. Published in Annals of Internal Medicine, this landmark research found no increased risk for autism, asthma, allergies, or autoimmune disorders among children receiving aluminum-containing vaccines. As healthcare professionals navigate persistent concerns about vaccine hesitancy, CIMA Care's evidence-based educational platform provides the scientific foundation needed to build vaccine confidence through comprehensive, WHO-aligned training and parent engagement strategies.
Aluminum Adjuvants: Modern Benefits and Mechanistic Evidence
Since the 1920s, when aluminum adjuvants were first shown to markedly enhance immune responses to diphtheria and tetanus toxoids, they have marked a pivotal milestone in vaccine development, laying the foundation for their modern role in ensuring potent, durable, and safe immunization.
Robust Immune Activation: Aluminum adjuvants are essential to maximizing vaccine effectiveness; modern studies confirm their potency, immunological benefits, and outstanding safety at current dosing levels.
Robust Immune Activation: 2025 immunology reviews indicate that aluminum adjuvants work by triggering an intricate cascade, promoting antigen adsorption, enhancing phagocytic uptake by antigen-presenting cells, and inducing the NLRP3 inflammasome, which results in the release of cytokines that recruit additional immune cells. [13]
Potentiation of Antibody Response: Recent experimental and clinical studies have confirmed that aluminum adjuvants reliably enhance antibody titers and induce long-lasting humoral immunity, especially for vaccines targeting extracellular bacteria and toxins. Furthermore, co-formulations (e.g., AS04 with monophosphoryl lipid A) have been shown to further augment T-cell and memory responses, offering superior protection compared to the antigen alone. [12]

Aluminum adjuvants activate immune cells for lasting protection.
The Largest Safety Study Ever Conducted Confirms Aluminum Adjuvant Safety
The Andersson et al. 2025 study represents the most comprehensive epidemiological investigation of aluminum vaccine adjuvants ever undertaken. Researchers analyzed 1,224,176 children born in Denmark between 1997 and 2018. Over this 21-year period, Denmark's vaccination schedule changed several times, which meant some children received more aluminum from their vaccines than others, ranging from zero to 4.5 milligrams in their first two years of life. This natural variation allowed scientists to compare health outcomes between children who received different amounts of aluminum.
The study examined 50 chronic disorders across three major categories: 36 autoimmune conditions, nine atopic/allergic disorders, and five neurodevelopmental conditions. Results showed no statistically significant increase in risk for any of the conditions studied.
What the researchers found when they compared children who received different amounts of aluminum from vaccines:
For every extra milligram of aluminum a child received, the study found:
- 2% fewer autoimmune diseases (like juvenile arthritis or celiac disease)
- 1% fewer allergic conditions (like asthma or food allergies)
- 7% fewer neurodevelopmental disorders (like autism or ADHD)
In other words, children who received more aluminum from vaccines actually had slightly lower rates of these conditions, not higher rates.
Why this matters for understanding vaccine safety:
The researchers employed advanced statistical methods to assess the confidence in their findings. Here is what they discovered:
- For 19 different health conditions, they could rule out even a slight 10% increase in risk from aluminum.
- For seven other conditions, they could rule out even a moderate 30% increase in risk.
In simple terms, this extensive study found no evidence that aluminum in vaccines causes health problems. In fact, the data showed the opposite pattern; children with higher aluminum exposure from vaccines were slightly healthier. However, this is likely due to other factors, such as better access to healthcare, rather than aluminum being protective.
The study's size (1.2 million children) and rigorous methods make this finding particularly reliable for assessing the safety of aluminum in vaccines. [1]
Expert Validation: Edward Belongia, a prominent vaccine safety scientist, described this as "the largest and most definitive observational study on the safety of vaccine-related aluminum exposure in children ever conducted." [2]

Each vaccine dose contains <1.5 mg aluminum. Danish cohort comparisons, enabled by exposure variations, found no evidence of added health risks.
Aluminum Adjuvants: Strong Safety Backed by Decades of International Evidence
WHO Endorsement: The World Health Organization's Global Advisory Committee on Vaccine Safety (GACVS) continues to support the safety of aluminum adjuvants based on comprehensive risk assessments spanning over 70 years of clinical use. The WHO's position emphasizes that the aluminum body burden never exceeds regulatory thresholds, even in vulnerable populations, including those with low birth weight. [3]
CDC Position: The U.S. Centers for Disease Control and Prevention maintains that aluminum salts have been used safely in vaccines for seven decades, with extensive surveillance data supporting continued use. The agency notes that aluminum adjuvants are essential for vaccine effectiveness, allowing reduced antigen requirements while enhancing immune responses. [4]
EMA Classification: Similarly, the European Medicines Agency classifies aluminum adjuvants as well-established compounds with demonstrated safety profiles across multiple vaccine formulations. [5]
Pharmacokinetic Insights: Recent pharmacokinetic research has refined the understanding of aluminum disposition in the body. Studies using isotopically labeled aluminum show that vaccine-derived aluminum is primarily deposited in bone tissue, with exposure levels remaining well below established safety thresholds. Infants receive approximately 4.4 milligrams of aluminum from vaccines in the first six months, compared to 7 milligrams from breast milk or 38 milligrams from formula feeding. [6 & 7]
FDA Review: According to the U.S. Food and Drug Administration, the quantity of aluminum salts used in approved vaccines is very small and rigorously tested; scientific studies confirm that the total aluminum exposure from all recommended vaccines in an infant’s first year poses an extremely low risk—remaining far below safety thresholds set by regulatory agencies, even for young or medically vulnerable children. [18]

WHO/FDA/CDC/EMA-approved aluminum adjuvants: 70+ years of proven safety in routine vaccines.
Addressing Persistent Vaccine Hesitancy Through Evidence-Based Education
Despite overwhelming scientific evidence supporting aluminum adjuvant safety, vaccine hesitancy related to these concerns continues to grow. Canadian surveillance data reveal that parents who are "really against" childhood vaccinations increased from 4% in 2019 to 17% in 2024, with aluminum-related concerns contributing to this fourfold rise. [8] This evidence-hesitancy gap underscores the critical need for improved communication strategies that bridge the disconnect between robust scientific findings and persistent public concerns about vaccine ingredients.
Healthcare providers report challenges in effectively communicating aluminum safety evidence, with over one-third feeling uncomfortable counseling vaccine-hesitant patients. Research identifies problematic communication approaches, including fear-based messaging and myth-busting strategies that may inadvertently increase familiarity with misinformation. [9]
Evidence-based communication recommendations emphasize empathetic listening, providing contextual information that compares vaccine aluminum to environmental exposures, transparent communication about safety data limitations, and personalized approaches tailored to specific concerns and educational levels. Healthcare provider training must incorporate updated talking points that reflect recent research findings while addressing concerns specific to aluminum that differ from those related to general vaccine hesitancy. [10]
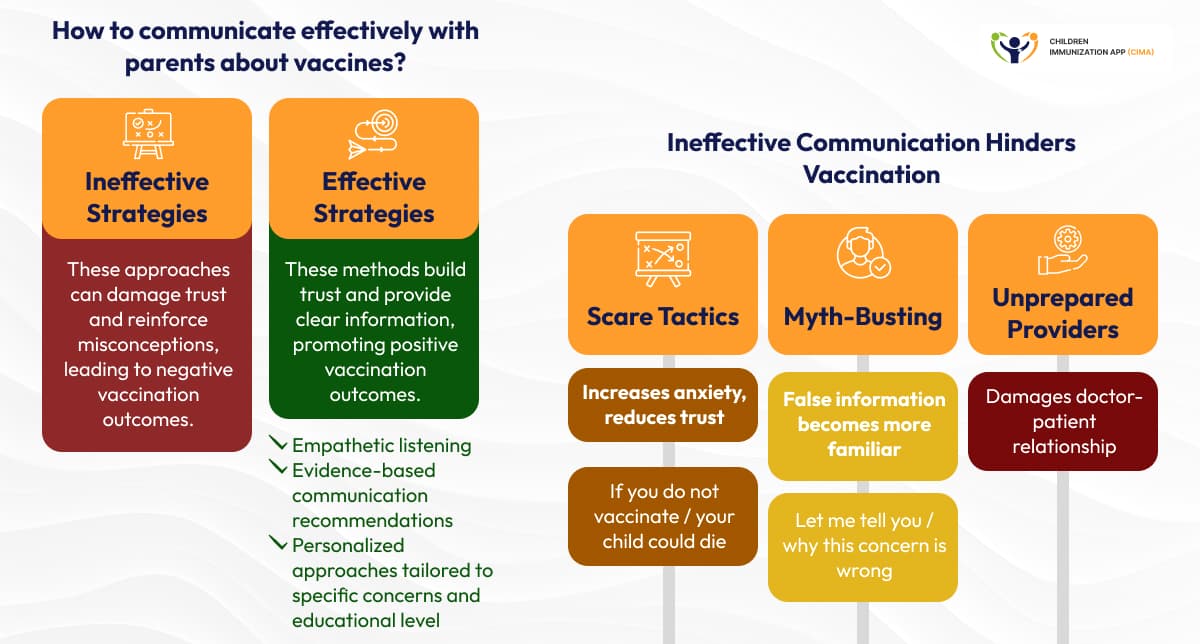
Empathetic, personalized, evidence-based communication outperforms scare and ineffective myth-busting in vaccine strategies.
Knowledge Gaps in Aluminum Adjuvant Research: Effectiveness and Biological Mechanisms
While aluminum adjuvants have been used in vaccines for over 70 years, recent systematic reviews reveal surprising limitations in our understanding of their effectiveness compared to placebo controls. Simultaneously, emerging research is uncovering new insights into how aluminum adjuvants work in the body while highlighting significant gaps in our knowledge about their long-term behavior and individual variability.
- Historical Knowledge Paradox: While aluminum adjuvants have been used in vaccines for over 70 years, recent systematic reviews reveal surprising limitations in our understanding of their effectiveness compared to placebo controls. Simultaneously, emerging research is uncovering new insights into how aluminum adjuvants work in the body while highlighting significant gaps in our knowledge about their long-term behavior and individual variability.
- Limited Effectiveness Evidence: The Russo Krauss et al. (2022) systematic review examined 102 randomized clinical trials involving 26,457 participants, comparing aluminum adjuvants with placebo or no intervention across all vaccine types. The experiment found little to no evidence of any difference between aluminum adjuvants and placebo or no intervention when measuring immune response, whether assessed by geometric mean antibody levels or by participants' rate of seroprotection. [11]
- Mechanistic Understanding Gaps: Research gaps persist, including limited data for special populations, individual variability in aluminum handling, and human long-term retention. Future priorities include extended follow-up, study of genetic influences on metabolism, and biomarker development for aluminum effects. [12]
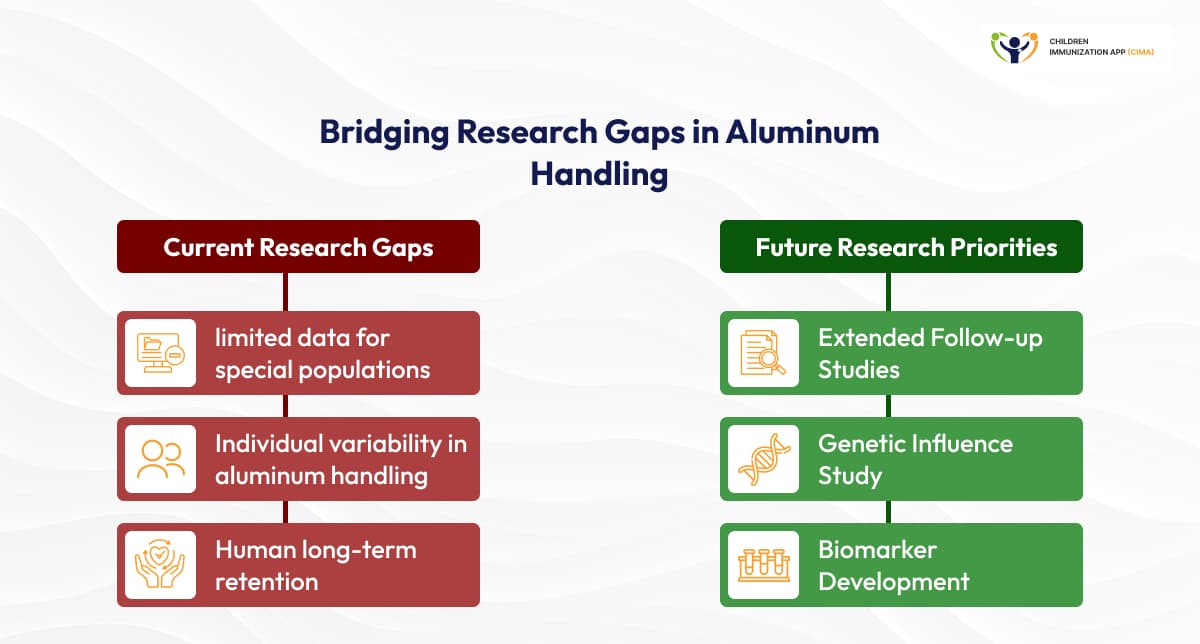
70+ years of use, yet notable gaps remain in aluminum handling and individual variability.
CIMA Care: Transforming Vaccine Confidence Through Scientific Excellence and Digital Innovation
CIMA Care's comprehensive ecosystem addresses the broad challenges of vaccine hesitancy and immunization gaps, among others, through interconnected approaches that align with the landmark Danish study findings and extensive international safety evidence. While CIMA Care's platform encompasses the full spectrum of vaccination science and public health education, the vaccine safety research reinforces the evidence-based foundation underlying all our educational initiatives and digital health solutions.
- Evidence-Based Healthcare Professional Education
The CIMA Health Academy empowers healthcare professionals with cutting-edge education through CPD-certified courses aligned with WHO, UNICEF, and international health authority guidelines.
- Strategic Parent and Community Engagement
The CIMA app delivers evidence-based safety information directly to parents through WHO-validated SMS messaging. Our communication strategy leverages findings from vaccine safety studies and international safety data to provide contextual safety information, milestone-based education, evidence-based and reliable myth-busting content, and insights into the effectiveness of positive communication strategies.
- Evidence-Based Publishing Initiative: CIMA Care implements the WHO's comprehensive framework for credible health information through strategic content creation across digital platforms. Our blog articles, published on both the CIMA Care website and LinkedIn, adhere to stringent credibility principles, featuring evidence-based content from current research, transparent source attribution, and clear disclosure of limitations. Each publication reflects the latest scientific understanding and helps healthcare enthusiasts acquire knowledge from reliable sources. Topics covered include vaccine safety evidence, global immunization challenges, and strategies for combating health misinformation and beyond.
- Real-World Impact and Global Reach
CIMA Care's evidence-based approach to vaccine safety communication has demonstrated measurable impact across diverse healthcare settings: Healthcare workers using CIMA Care's platform report that the Academy's evidence-based safety education enhances their ability to address parent concerns while enabling confident, science-based discussions about vaccine ingredients. Parents and healthcare workers describe our safety messaging as providing clear and understandable information that alleviates concerns about vaccine components. [14-17] For more information on the feedback from the Academy's participants, nurses, and parents, please visit:
1- Healthcare CPD Stories
2- CIMA Jordan & CDC Collaboration Success Stories
3- CIMA: Revolution in Child Health
4- Digital Innovation at Atuakom

CIMA's evidence-based articles and participant testimonials: global impact across 77+ countries.
Future-Ready Aluminum Safety Communication
CIMA Care's platform continuously evolves to incorporate the latest vaccine safety research, ensuring healthcare professionals and parents receive up-to-date information based on the strongest available evidence. Our commitment to scientific excellence positions CIMA Care as the premier resource for evidence-based vaccine safety education worldwide.
Through accessible, interactive, and practical training modules, CIMA Care equips healthcare professionals to become confident vaccine safety advocates while empowering parents with accurate, WHO-validated information. By addressing both the technical and communication challenges surrounding vaccine safety, CIMA Care builds the foundation for sustained vaccine confidence and optimal child health outcomes globally.

CIMA Care’s dual platform builds vaccine confidence: the app and health messages for families and the academy for professionals.
Image References
1- Adobe Stock. Doctor hand holds syringe and vaccine [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/doctor-hand-holds-syringe-and-vaccine/199775698
2- Adobe Stock. Automated pharmaceutical machinery filling glass vaccine vials with aluminum caps [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/automated-pharmaceutical-machinery-filling-glass-vaccine-vials-with-aluminum-caps-medical-manufacturing-equipment-for-drug-production-and-packaging-healthcare-biotechnology-medicine-industry/1612030115
3- Adobe Stock. Parents carrying son on shoulders on beach vacation [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/parents-carrying-son-on-shoulders-on-beach-vacation/246367828
- 4- Adobe Stock. Portrait of an African American woman and her family [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/portrait-of-an-african-american-woman-and-her-family/314803619
- 5- Adobe Stock. Young African American woman holding her baby’s hand at home [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/young-african-american-woman-holding-her-baby-s-hand-at-home/543224844
- 6- OZ Biosciences. AlumVax Hydroxide [Internet]. Marseille (FR): OZ Biosciences SAS; [cited 2025 Oct 5]. Available from: https://ozbiosciences.com/aluminum-gels/79-alumvax-hydroxide.html
Blog Resources
- 1- Andersson, N.W., Svalgaard, I.B., Hoffmann, S.S., & Hviid, A. (2025). Aluminum-Adsorbed Vaccines and Chronic Diseases in Childhood: A Nationwide Cohort Study. Annals of Internal Medicine. Aluminum-Adsorbed Vaccines and Chronic Diseases in Childhood: A Nationwide Cohort Study: Annals of Internal Medicine: Vol 0, No 0
- 2- Major new study finds no health risks from aluminium in childhood vaccines [Internet]. Gavi, the Vaccine Alliance. 2025 [cited 2025 Aug 27]. Available from: Major new study finds no health risks from aluminium in childhood vaccines
- 4- CDC. Adjuvants and Vaccines [Internet]. Vaccine Safety. 2024. Available from: Adjuvants and Vaccines
- 5- Adjuvants in vaccines for human use - Scientific guideline | European Medicines Agency (EMA) [Internet]. European Medicines Agency (EMA). 2005 [cit S ed 2025 Aug 27]. Available from: Adjuvants in vaccines for human use - Scientific guideline | European Medicines Agency (EMA)
- 6- Vaccine Ingredients: Aluminum [Internet]. Welcome | Children's Hospital of Philadelphia. 2022. Available from: Vaccine Ingredients: Aluminum
- 7- Mitkus RJ, King DB, Hess MA, Forshee RA, Walderhaug MO. Updated aluminum pharmacokinetics following infant exposures through diet and vaccination. Vaccine [Internet]. 2011 Nov 28;29(51):9538–43. Available from: NCBI - WWW Error Blocked Diagnostic
- 8- Korzinski D. Parental opposition to childhood vaccination grows as Canadians worry about harms of anti-vax movement [Internet]. Angus Reid Institute. 2024. Available from: Parental opposition to childhood vaccination grows as Canadians worry about harms of anti-vax movement - Angus Reid Institute
- 9- Löffler P. Review: Vaccine Myth-Buster – Cleaning Up With Prejudices and Dangerous Misinformation. Frontiers in Immunology. 2021 Jun 10;12.
- 10- Aluminum Adjuvants Talking Points [Internet]. Association of Immunization Managers. 2022. Available from: Aluminum Adjuvants Talking Points
- 11- Krauss SR, Barbateskovic M, Klingenberg SL, Djurisic S, Petersen SB, Kenfelt M, et al. Aluminium adjuvants versus placebo or no intervention in vaccine randomised clinical trials: a systematic review with meta-analysis and Trial Sequential Analysis. BMJ Open. 2022 Jun;12(6):e058795.
- 12- Francisco H. News-Medical [Internet]. News-Medical. 2025 [cited 2025 Aug 28]. Available from: Aluminum in Vaccines: Mechanisms, Myths, and Safety Data
- 13- Xing J, Zhao X, Li X, Fang R, Sun M, Zhang Y, et al. The recent advances in vaccine adjuvants. Frontiers in Immunology. 2025 May 13;16.
- 14- Cima Children Immunization app - Voices & Stories [Internet]. CIMA Care. 2025. Available from: Cima Children Immunization app - Voices & Stories
- 15- Cima Children Immunization app - Voices & Stories [Internet]. CIMA Care. 2025 [cited 2025 Aug 28]. Available from: Cima Children Immunization app - Voices & Stories
- 16- CIMA | Children Immunization App [Internet]. Cima.care. 2025. Available from: Cima Children Immunization app - Voices & Stories
- 17- Cima Children Immunization app - Voices & Stories [Internet]. CIMA Care. 2025. Available from: Cima Children Immunization app - Voices & Stories
- 18- Center for Biologics Evaluation and Research. Common Ingredients in FDA-Approved Vaccines. FDA [Internet]. 2024 Jan 12; Available from: Common Ingredients in FDA-Approved Vaccines
Enjoyed this article?
Share it with your friends on LinkedIn: Conclusive Evidence: Addressing Concerns on Aluminum Vaccine Adjuvant Safety\
Follow us on LinkedIn for more updates and insights: Cima Care GmbH
