Robust Vaccine Safety: Cutting-Edge Science & Worldwide Monitoring |
October 20, 2025 . 9 Minutes read
Vaccine Safety: Evidence, Controversies, and Communication
The landscape of vaccine safety continues to evolve rapidly, with robust scientific evidence supporting the safety profiles of vaccines. In contrast, ongoing misinformation and legitimate safety questions require nuanced, evidence-based responses from healthcare professionals. While vaccine hesitancy continues, the facts remain powerful: serious complications remain extraordinarily rare, cutting-edge monitoring systems catch safety signals faster than ever before, and healthcare providers who master evidence-based communication consistently overcome patient doubts.
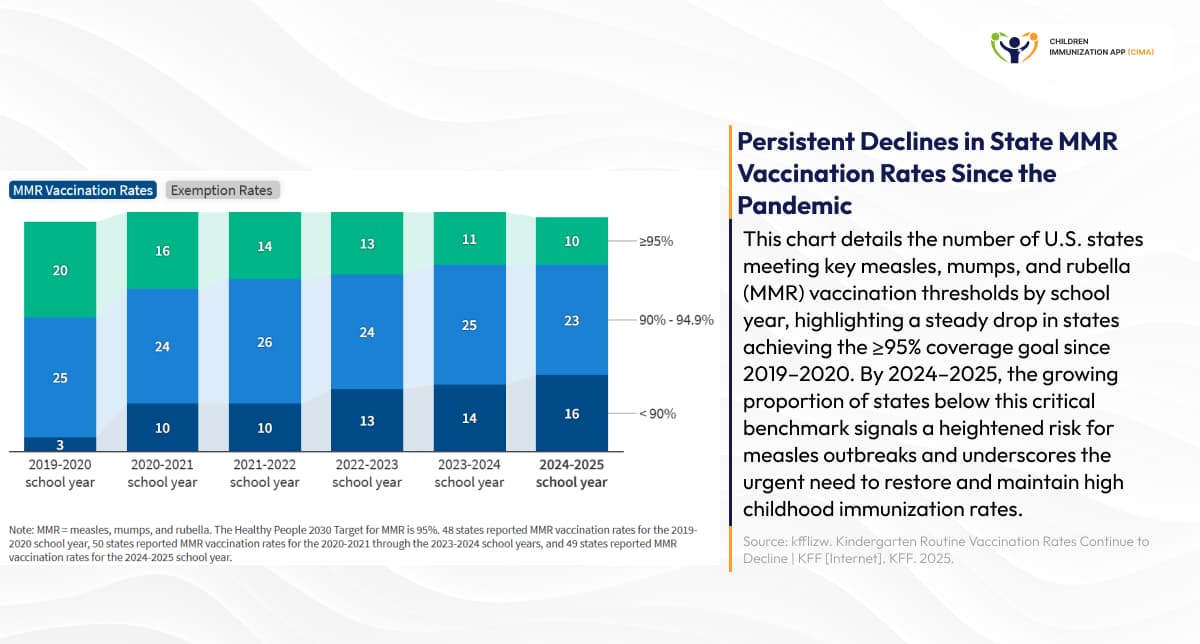
Stark reality: Fewer states achieving 95% MMR vaccination goal, heightening outbreak risk.
The Provider Challenge: Most Trusted Source, Toughest Conversations
Healthcare providers see themselves as the most trusted source of vaccine information, [4] making their communication skills critical to addressing both legitimate safety concerns and pervasive misinformation. The challenge lies in balancing acknowledgment of rare but real adverse events with clear communication about overwhelming risk-benefit ratios favoring vaccination. [5]
While vaccine hesitancy continues, the facts remain powerful: serious complications remain extraordinarily rare, cutting-edge monitoring systems catch safety signals faster than ever before, and healthcare providers who master evidence-based communication consistently overcome patient doubts. [1]
Evidence-based communication strategies show measurable effectiveness in addressing hesitancy. Clinics that trained their healthcare workers with a multi-component approach, including presumptive recommendations, motivational interviewing, fact sheets, and other tactics, had ~9.5% higher HPV vaccine initiation than clinics that did not. Training healthcare providers in motivational interviewing techniques and patient-centered shared decision-making significantly improves provider confidence across multiple domains. [33]

Multilevel communication strategies help providers overcome vaccine hesitancy.
Landmark safety evidence from 2024-2025 multinational studies
The most comprehensive vaccine safety studies in history have emerged from 2024-2025, analyzing data from nearly 100 million vaccinated individuals across multiple countries and platforms. These unprecedented investigations provide definitive answers to persistent safety questions while quantifying rare adverse events with remarkable statistical precision. From long-term mRNA vaccine follow-up studies to authoritative National Academies reviews, the evidence demonstrates robust safety profiles across all age groups and populations. The scale and rigor of these multinational analyses establish a new standard for vaccine safety assessment, offering healthcare providers concrete data to address patient concerns with confidence.
- mRNA COVID-19 vaccine: Recent peer-reviewed studies provide substantial evidence for the long-term safety of the mRNA COVID-19 vaccine. The COVE trial long-term follow-up published in Nature Communications (2024) tracked 29,035 participants through April 2023, with a median follow-up exceeding 25 months post-primary vaccination. No new safety concerns emerged during extended monitoring, with serious vaccine-related adverse events occurring in fewer than 0.1% of booster recipients. [6] Critically, immunocompromised patients demonstrated similar or lower adverse event rates compared to healthy controls, supporting vaccination in vulnerable populations. [7]
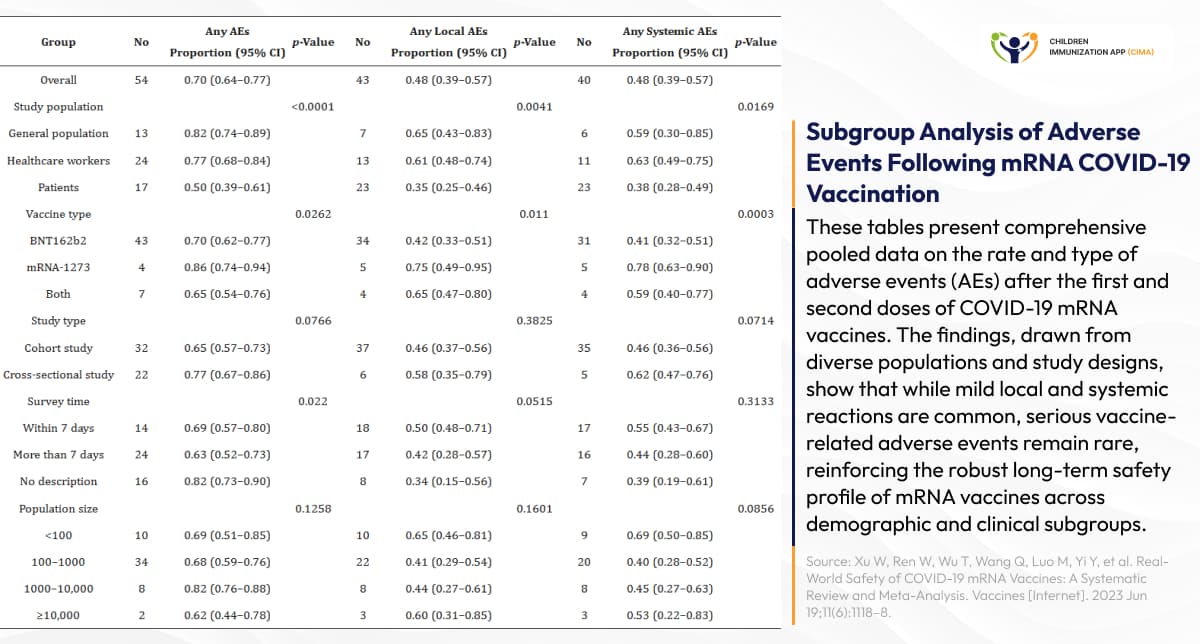
Comprehensive data shows rare adverse events across mRNA COVID-19 vaccine populations.
- mRNA Vaccines and Myocarditis and Pericarditis: Myocarditis is the inflammation of the heart muscle, which can weaken the heart's pumping ability and cause chest pain or irregular heartbeats, while pericarditis is the inflammation of the thin sac around the heart, often causing sharp chest pain that worsens with deep breaths or when lying down. [8] FDA data for the 2023–2024 mRNA COVID-19 vaccines indicate an estimated rate of about 8 cases of myocarditis or pericarditis per million doses in individuals aged 6 months through 64 years, and approximately 27 cases per million doses in males aged 12 through 24 years within one week of vaccination. [9] However, recent cardiac MRI follow-up studies of approximately 300 patients with vaccine-associated myocarditis revealed persistent abnormal findings at 5-month follow-up in some patients, with unknown clinical significance. [10] The CDC's Vaccine Safety Datalink found no statistically significant signal for myocarditis with 2023-2024 mRNA vaccines, and 80% of diagnosed patients achieved full or probable full recovery according to their cardiologists. [11] Leading experts, including Dr. Paul Offit from the Children's Hospital of Philadelphia, note that vaccine-associated myocarditis cases have declined since early vaccine rollout, stating, "We don't see it so much anymore" due to widespread vaccination and natural immunity. [12] Notably, for young men aged 18–25, there is an excess risk of 22.4 myocarditis cases per million mRNA-BNT162b2 (Pfizer) doses and 31.2 per million mRNA-1273 (Moderna) doses in the week after vaccination. To compare the risk of myocarditis after SARS-CoV-2 infection, the risk is approximately 150 per 100,000 in US military men aged 20–39, which is quite higher than the vaccine risk. As such, the risk-benefit calculation remains favorable. [13]

FDA data: Myocarditis is rare after mRNA vaccines, with 83-90% recovery rates documented.
- The Global COVID-19 Vaccine Safety (GCoVS) Project: Published in the Vaccine journal, this paper represents the largest vaccine safety analysis ever performed, encompassing 99,068,901 vaccinated individuals across 10 sites in 8 countries. This multinational study analyzed 183,559,462 BNT162b2, 36,178,442 mRNA-1273, and 23,093,399 ChAdOx1 (non-replicating adenoviral vector vaccine) doses with 23,168,335 person-years of follow-up, providing unprecedented statistical power to detect rare adverse events. Key statistical findings confirmed previously identified safety signals while quantifying risks with remarkable precision. Observed-to-expected ratios with the lower bound of the confidence interval >1.5 were identified for Guillain-Barré syndrome (2.49, 95% CI: 2.15–2.87) after the ChAdOx1 first dose; cerebral venous sinus thrombosis (3.23, 95% CI: 2.51–4.09) after the ChAdOx1 first dose; and acute disseminated encephalomyelitis (3.78, 95% CI: 1.52–7.78) after the mRNA-1273 (Moderna, Spikevax) first dose. As noted by GVDN Co-Director Dr. Helen Petousis-Harris, “By making the data dashboards publicly available, we are able to support greater transparency and stronger communications to the health sector and public.”
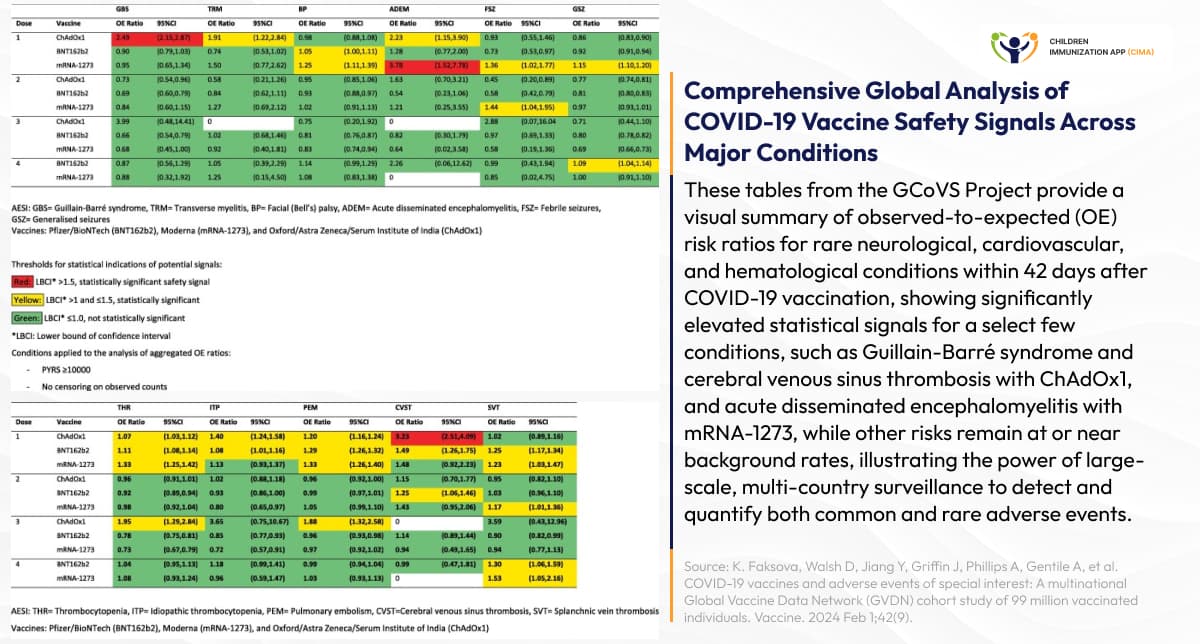
Global vaccine safety analysis covers 99 million vaccinated individuals and major conditions.
- National Academies of Sciences, Engineering, and Medicine (NASEM) Review: The National Academies Evidence Review, published in April 2024, provided an authoritative scientific consensus. The National Academies of Sciences committee concluded that Pfizer and Moderna vaccines can cause myocarditis but do not cause infertility, Guillain-Barré syndrome, Bell's palsy, thrombosis with thrombocytopenia syndrome, or heart attack. Committee Vice Chair Anne Bass emphasized, "Despite a large body of evidence from extraordinary efforts by investigators around the world, our committee found that in many cases, if not most, evidence was insufficient to accept or reject causality for a particular potential harm from a specific COVID-19 vaccine."
- Pediatric Safety Data: The safety data from 2024–2025 provides exceptional reassurance for parents and providers. A Nature Communications study of 5.1 million children in England found no increased risks of adverse events 1–42 days following COVID-19 vaccination in children ages 5–11.

Authoritative review: Evidence insufficient to confirm most COVID vaccine harm claims.
Specific safety concerns receive detailed scientific investigation
Beyond broad population studies, researchers have conducted targeted investigations into the most frequently questioned vaccine safety concerns, providing definitive answers to persistent doubts. Large-scale cohort studies involving over 1.2 million children have addressed aluminum adjuvant safety, while comprehensive analyses confirm vaccination safety during pregnancy and for children with genetic disorders or diabetes. These focused investigations transform abstract safety concerns into concrete, evidence-based conclusions that directly address the specific worries parents and healthcare providers encounter daily. The depth of investigation into these particular areas demonstrates the scientific community's commitment to leaving no safety question unanswered.
- Aluminum adjuvant safety evidence: A landmark Danish cohort study analyzing 1.2 million children over 24 years provides conclusive evidence that aluminum adjuvants in childhood vaccines pose no increased risk for autism, asthma, allergies, or autoimmune disorders, representing the largest and most definitive observational study on vaccine-related aluminum safety ever conducted. Despite this robust safety evidence supported by seven decades of international regulatory approval, vaccine hesitancy related to aluminum concerns has quadrupled among Canadian parents (4% to 17% between 2019–2024), revealing critical gaps between scientific evidence and public perception. The research paradoxically shows surprising limitations in effectiveness data, with systematic reviews demonstrating little evidence of immune response differences between aluminum adjuvants and placebo controls despite 70+ years of clinical use. Evidence-based communication strategies emphasizing empathetic engagement and contextual safety information are essential for healthcare providers to effectively address persistent aluminum-related vaccine concerns and rebuild public confidence in immunization programs.

Landmark study of 1.2M children confirms aluminum adjuvants are safe in childhood vaccines.
- Pregnancy vaccination: The safety data provide strong evidence for maternal and neonatal benefits. Maternal vaccination during pregnancy provides dual protection for mothers and infants through efficient transplacental antibody transfer via the neonatal Fc receptor (FcRn), with optimal transfer occurring during the third trimester and varying by vaccine timing and platform. Robust evidence from large-scale studies supports the safety and effectiveness of four recommended vaccines (COVID-19, influenza, Tdap, and RSV), with no increased risks of adverse pregnancy outcomes and significant protection against serious infant diseases, including 81.8% efficacy for RSV and 80% effectiveness for late-pregnancy COVID-19 vaccination. Despite overwhelming scientific consensus from the WHO, CDC, and ACOG, vaccine hesitancy during pregnancy persists, with provider communication strategies emphasizing presumptive recommendations proving most effective in achieving vaccination rates of 61.4% versus 22.7% without recommendations. The maternal vaccination landscape continues expanding with promising vaccines in development for Group B Streptococcus, cytomegalovirus, and other perinatal pathogens, potentially preventing tens of thousands of infant deaths and complications globally while addressing current gaps in protection against major pregnancy-related infections.

Dual protection: Maternal vaccination safely shields both mother and baby during pregnancy.
- Type 1 Diabetes and Vaccination: Vaccine safety for children with Type 1 Diabetes (T1D) is well-established, with multiple large-scale epidemiological studies of over 739,694 children confirming no causal relationship between routine childhood immunizations and T1D development, while demonstrating that children with diabetes require all standard vaccinations due to their threefold higher risk of severe infections. Emerging evidence reveals potential protective benefits, including a 13% reduction in T1D risk associated with rotavirus vaccination through prevention of molecular mimicry mechanisms, and promising therapeutic effects from BCG vaccination showing sustained 10% reductions in HbA1c levels through immune cell metabolic reprogramming. Current scientific consensus emphasizes that vaccination remains not only safe but essential for T1D patients, with ongoing research exploring vaccines as both prevention and treatment modalities rather than risk factors for this autoimmune condition.
Large studies confirm vaccines are safe for Type 1 diabetes patients, showing therapeutic promise.
- Genetic Vulnerabilities and Vaccination: Vaccine safety for children with genetic disorders is well-established through extensive clinical evidence demonstrating that standard immunization schedules remain safe and essential for this vulnerable population, with modifications required primarily for severely immunocompromised children or those receiving high-dose corticosteroid therapy who may need timing adjustments for live vaccines. Current research reveals that children with genetic conditions face significantly higher risks from vaccine-preventable diseases than from vaccination itself, with studies across metabolic disorders, chromosomal conditions, neuromuscular diseases, and connective tissue disorders consistently showing normal vaccine responses and safety profiles when appropriate protocols are followed. Despite overwhelming scientific support for vaccination in genetic disorders, concerning coverage gaps persist due to healthcare provider uncertainty and parental hesitancy, highlighting the critical need for individualized risk-benefit assessments, specialized clinical protocols, and enhanced education to ensure these medically vulnerable children receive optimal protection through evidence-based immunization strategies.

Research proves vaccines safe for children with genetic conditions, addressing provider fears.
Revolutionary advances and AI-powered safety surveillance systems
Vaccine safety monitoring has been transformed by artificial intelligence and machine learning technologies that process millions of reports in real-time, detecting safety signals faster and more accurately than ever before. The FDA's BEST (Biologics Effectiveness and Safety) system, WHO's AI-powered early warning networks, and sophisticated machine learning algorithms have revolutionized pharmacovigilance from reactive monitoring to proactive risk prediction. These cutting-edge systems achieve remarkable performance improvements, reducing manual review workloads by over 80% while maintaining 100% capture rates of relevant safety signals. The integration of social media monitoring, automated literature reviews, and predictive analytics creates an unprecedented safety surveillance infrastructure that protects public health through continuous innovation.
- Artificial Intelligence: It has transformed vaccine safety monitoring from reactive surveillance to proactive risk prediction, enabling the early detection of adverse events and the rapid implementation of protocol updates to protect public safety.
- Real-time Monitoring Systems: These systems now provide unprecedented surveillance capabilities. The FDA's BEST (Biologics Effectiveness and Safety) System enables rapid queries across millions of vaccinated individuals through partnerships with IBM Federal HealthCare, IQVIA, and Columbia University. Near real-time safety surveillance successfully identified myocarditis/pericarditis and anaphylaxis signals consistent with published literature while monitoring 17 specific adverse events of special interest.
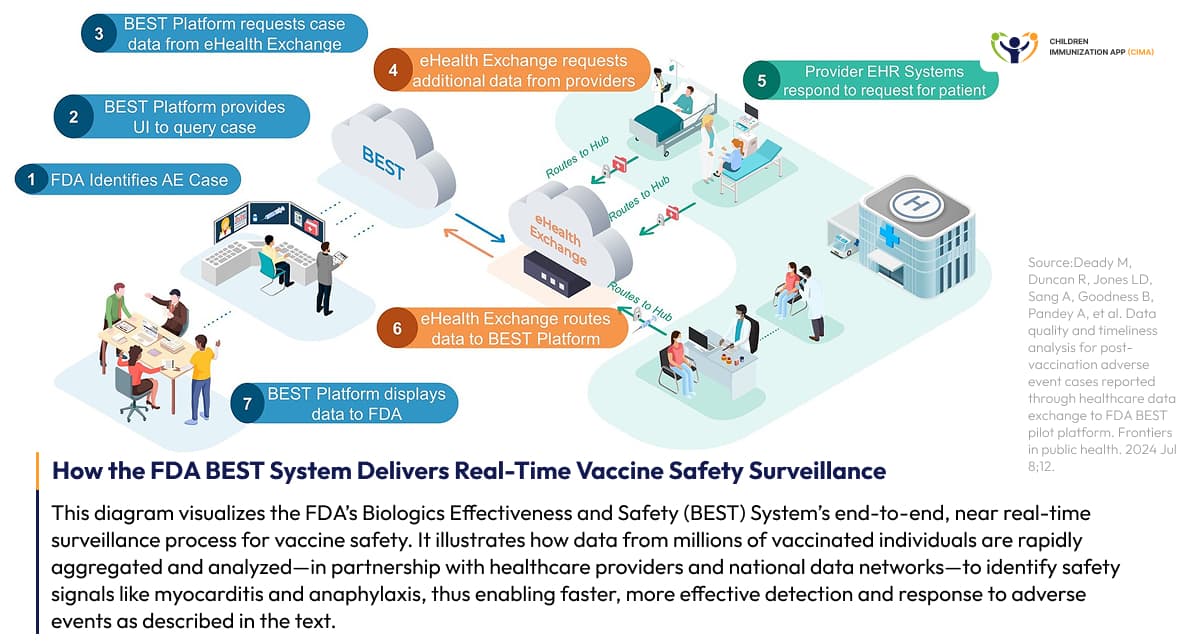
AI-powered Biologics Effectiveness and Safety, or BEST, system transforms vaccine safety with real-time surveillance technology.
- Machine learning algorithms: Such algorithms demonstrate remarkable performance improvements in pharmacovigilance efficiency. Literature review automation has reduced the volume of human reviews by over 80% while maintaining a 100% capture rate of relevant safety papers. Causality assessment models achieve an AUC score of 0.924 (95% CI, 0.922–0.927) with a sensitivity of 0.900 and a positive predictive value of 0.778, substantially improving upon traditional manual assessments. AUC measures how well a classification model can distinguish between two classes (e.g., adverse event vs. no event). An AUC of 1.0 indicates a perfect model, while 0.5 suggests the model performs no better than random guessing.
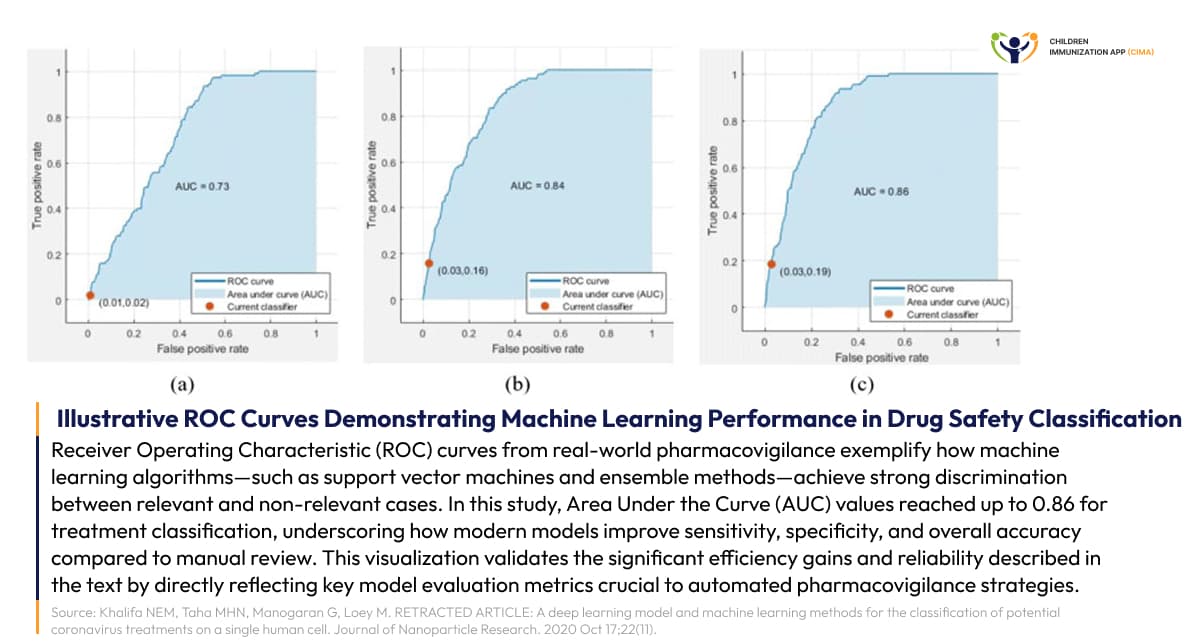
Receiver Operating Characteristic, or ROC, curves demonstrate machine learning achieving 80%+ efficiency in drug safety review. Area Under the Curve, or AUC, summarizes performance.
- WHO Early Warning System: This system utilizes AI-powered content analysis from social media and online sources to provide real-time monitoring of vaccine-related concerns and misinformation. This complements functional vaccine safety surveillance systems by identifying emerging safety perceptions before they impact vaccination behavior, enabling proactive public health responses.
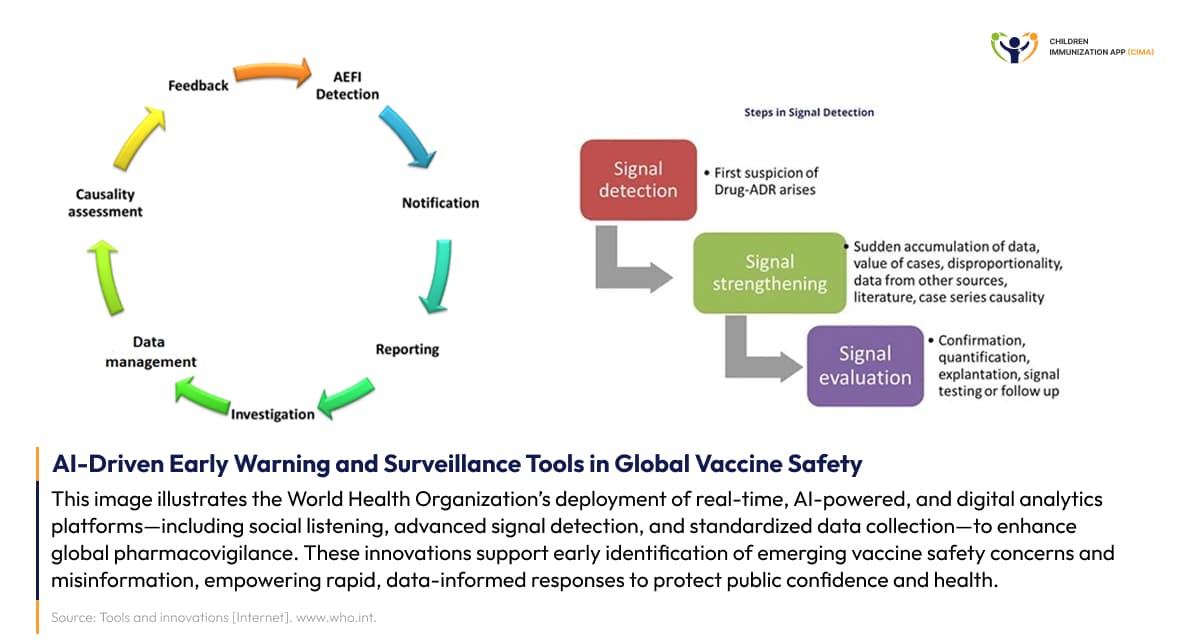
WHO deploys AI-powered early warning system for proactive global vaccine safety monitoring.
International Collaboration Reaches Unprecedented Levels of Coordination.
Global vaccine safety efforts have achieved remarkable coordination through standardized reporting systems, international coalitions, and harmonized case definitions, enabling seamless data sharing across continents. The WHO's case-based reporting standards now connect 92 countries in systematic adverse event monitoring, while organizations like ICMRA and the Brighton Collaboration ensure consistent, scientifically rigorous approaches to safety assessment worldwide. Joint regulatory initiatives, collaborative workshops, and unified communication strategies have created an integrated global response system that rapidly addresses emerging safety concerns. This unprecedented level of international cooperation ensures that vaccine safety insights discovered anywhere in the world are immediately applied to global public health protection efforts.
- Global vaccine safety surveillance: This surveillance has achieved remarkable standardization and coordination in recent years. The WHO's new case-based reporting standard shows that in 2022, 92 countries (43% of 215 WHO countries) achieved the target of at least one serious AEFI per 1 million population annually, representing significant progress from 51 countries (24%) in 2020. Joint reporting between National Regulatory Authorities and Expanded Programs on Immunization increased from 19% to 27% of countries.
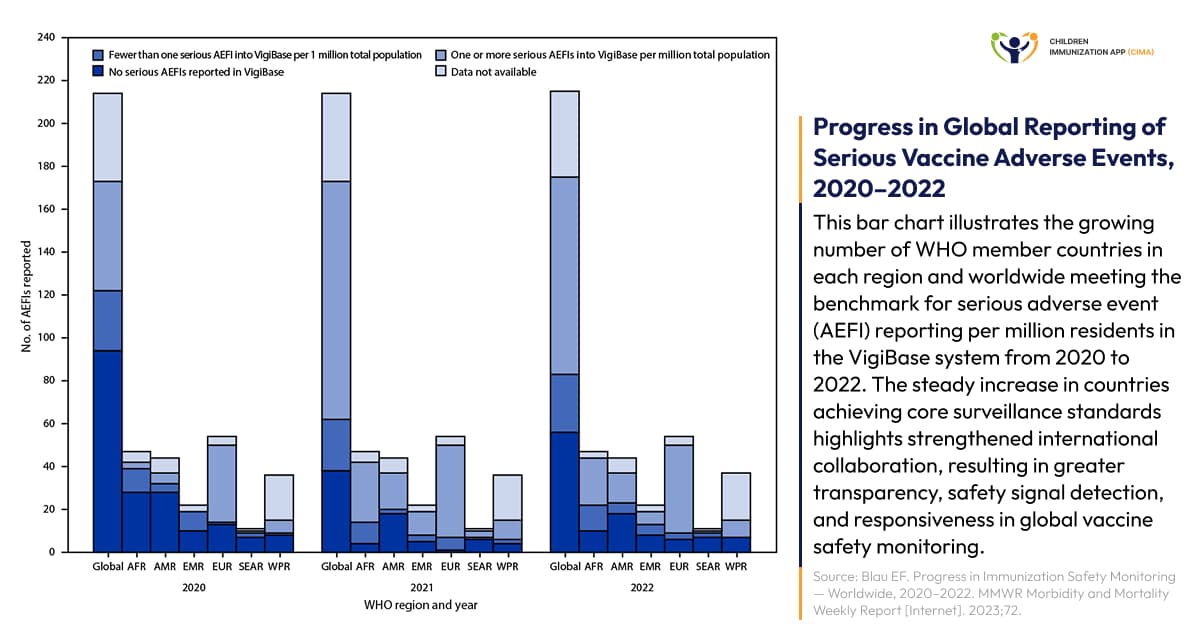
Global progress: 92 WHO countries met vaccine safety reporting standards in 2022, up from 51 in 2020.
- The International Coalition of Medicines Regulatory Authorities (ICMRA): ICMRA facilitates active collaboration among medicines regulators worldwide from all global regions. Through collaborative workshops and the Vaccine Pharmacovigilance Network, ICMRA enables proactive information sharing, coordination of safety analyses, and signal detection related to COVID-19 vaccines. A January 2021 workshop focused on practical steps for joint active and passive vaccine safety monitoring, exchanging emerging safety signals, simulation exercises, and consistent global communication strategies, all aimed at reinforcing public vaccine confidence. Joint ICMRA and WHO initiatives leverage regional and global networks to ensure rapid collaboration, evaluation of adverse events of special interest, and harmonized scientific review of COVID-19 vaccines and safety surveillance.

The International Coalition of Medicines Regulatory Authorities, or ICMRA, shows positive collaboration impact.
- The Brighton Collaboration: The Brighton Collaboration is an international network that develops standardized definitions and guidelines for identifying and reporting vaccine safety issues, enabling health professionals worldwide to recognize side effects after vaccination using clear criteria reliably. The Brighton Collaboration's standards aim to establish uniform case definitions for vaccine adverse events, facilitating easier monitoring and comparison of vaccine safety data for researchers and health organizations. Case definitions, such as for Thrombosis with Thrombocytopenia Syndrome (TTS) and Vaccine-Induced Immune Thrombocytopenia and Thrombosis (VITT), are detailed descriptions that explain exactly what signs, symptoms, and laboratory tests are needed to identify and confirm these conditions after a vaccination. These definitions are regularly updated as new information becomes available, and new definitions, such as for autoimmune hepatitis, have been introduced to address important safety concerns highlighted by recent vaccine experiences.

Global standards: Brighton case definitions unite vaccine safety professionals’ reports worldwide.
Conclusion
The vaccine safety landscape in 2024-2025 demonstrates unprecedented scientific rigor, technological innovation, and international cooperation. [39] Multinational studies involving nearly 100 million individuals provide definitive evidence of vaccine safety while quantifying rare adverse events with remarkable precision. [40] AI-powered surveillance systems process millions of reports in real-time, enabling proactive risk detection and rapid response to emerging signals. [41]
Yet significant challenges remain: declining childhood vaccination rates threaten population immunity, misinformation spreads faster than interventions, and complex safety communications require evidence-based approaches.[31] The path forward requires continued investment in innovative monitoring technologies, evidence-based communication strategies, and international collaboration while maintaining an unwavering commitment to scientific integrity and public transparency.
The evidence overwhelmingly supports continued confidence in vaccine safety based on the most comprehensive surveillance infrastructure ever developed. As we advance into an era of personalized, predictive safety monitoring, the foundation established in 2024-2025 will ensure vaccines remain among the safest and most effective public health interventions available, protecting individuals and communities from vaccine-preventable diseases while maintaining the highest safety standards through continuous innovation and vigilance.[39]

Five pillars of modern vaccine safety create comprehensive framework for public protection.
CIMA Care: Transforming Vaccine Safety Communication Through Digital Innovation and Evidence-Based Education
As vaccination rates decline despite unprecedented safety evidence from AI-powered surveillance and multinational studies, the critical gap lies in translating robust science into trusted communication that reaches healthcare providers and families when decisions matter most. Yet as misinformation spreads faster than interventions, the critical gap lies not in safety data but in translating this evidence into trusted, accessible communication.
CIMA Care's integrated digital health ecosystem directly addresses the core challenges identified in this vaccine safety landscape through three interconnected approaches that bridge the evidence-to-practice gap:
- Evidence-Based Healthcare Professional Education: The CIMA Health Academy empowers healthcare professionals with cutting-edge vaccine safety communication training through CPD-recognized courses that have, so far, reached over 2,762 practitioners across 77+ countries. Our curriculum integrates the latest safety evidence with proven communication strategies that transform provider confidence in addressing vaccine hesitancy. Through our courses and blog articles, healthcare workers can have an enhanced ability to discuss complex safety topics, from myocarditis risk-benefit ratios to aluminum adjuvant safety, using the same evidence-based frameworks that inform global regulatory decisions.

Digital innovation: CIMA Care transforms vaccine safety communication for, so far, 2,762+ providers by supporting them to develop their proficiency.
- Safety Monitoring and Response: CIMA Care's upcoming Adverse Event Following Immunization (AEFI) monitoring system provides healthcare providers with sophisticated tools for identifying, documenting, and analyzing safety signals at both clinic and population levels. Our hierarchical data visualization, advanced filtering capabilities, and comprehensive reporting tools enable the same proactive safety monitoring that has revolutionized global vaccine surveillance, while ensuring that safety concerns are addressed with the transparency and responsiveness that builds public confidence.

CIMA Care's Adverse Event Following Immunization system enables vaccine safety monitoring.

Trusted communication: CIMA Care delivers WHO/UNICEF-validated vaccine info through SMS health messages.
- Strategic Parent and Community Engagement: Through WHO-validated SMS messaging and multilingual health education, CIMA Care delivers the same caliber of evidence-based safety information highlighted in this article directly to parents during critical decision-making moments. Our automated communication system addresses vaccine safety concerns using content validated by international health authorities, providing contextual risk information and myth-busting content that counters the misinformation contributing to declining vaccination rates documented in this analysis.
- Proven Real-World Impact: CIMA Care's evidence-based approach has demonstrated measurable success in addressing the outlined vaccination challenges, with documented improvements of 19% in Jordan's Zaatari Refugee Camp and 23% in Cameroon's Bamenda Region. These outcomes validate the effectiveness of combining robust safety monitoring with trusted communication channels, the same combination identified as essential for maintaining public confidence in the evolving vaccine safety landscape.

Measurable success: CIMA Care's trusted approach improves vaccination rates 19-23% globally.

Path forward: CIMA Care helps in transforming vaccine safety evidence into confident healthcare action through knowledge development and reliable research-based finding dissemination.
The Path Forward: Transforming Evidence into Action
As the international vaccine safety community continues advancing through transnational surveillance, standardized global reporting, and unprecedented scientific collaboration, CIMA Care stands ready to bridge the critical last mile between cutting-edge safety evidence and confident, informed vaccination decisions. Our comprehensive platform ensures that the remarkable vaccine safety achievements documented in 2024-2025 translate into sustained public trust and optimal immunization coverage worldwide.
Join CIMA Care in transforming vaccine safety communication from a reactive response to proactive confidence-building. Visit www.cima.care to discover how our evidence-based digital solutions can strengthen your vaccine safety monitoring and communication capabilities, ensuring every child receives life-saving protection through informed, confident healthcare decisions.
In the digital age of vaccine safety, robust surveillance systems provide the foundation, but trusted communication builds the bridge to public confidence.
Image References
1. Adobe Stock. Covid 19 realtime data on mobile application. Hands of a woman checking overview worldwide Coronavirus disease cases report while travelling [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/covid-19-realtime-data-on-mobile-application-hands-of-a-woman-checking-on-overview-worldwide-coronavirus-disease-cases-report-while-travelling-vaccinations-herd-immunity-safety-transportation/430910992
2. Adobe Stock. An open refrigerator stocked with multiple blue-capped vaccine vials representing healthcare and medical storage [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/an-open-refrigerator-stocked-with-multiple-blue-capped-vaccine-vials-representing-healthcare-and-medical-storage/772354065
3. Adobe Stock. Doctor adjusts the syringe to inject the vaccine to an African child, concept of COVID-19 vaccination in Africa [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/doctor-adjusts-the-syringe-to-inject-the-vaccine-to-an-african-child-concept-of-covid-19-vaccination-in-africa-coronavirus-vaccine/487670345
4. Adobe Stock. Development and manufacture process of a new vaccine, final production of filled vials of COVID-19 vaccine [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/development-and-manufacture-process-of-a-new-vaccine-final-production-of-filled-vials-of-covid-19-vaccine-bio-science-3d-illustration/410157556
5. Adobe Stock. A close-up image of a medical syringe highlighting its needle and fluid chamber against a plain background [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/a-close-up-image-of-a-medical-syringe-highlighting-its-needle-and-fluid-chamber-against-a-plain-background/1408725336
6. Adobe Stock. Pharmaceutical capsules technology global health smart AI-driven vaccine capsules enabling needle-free immunization for mass vaccination efforts [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/pharmaceutical-capsules-technology-global-health-smart-ai-driven-vaccine-capsules-enabling-needle-free-immunization-for-mass-vaccination-efforts/1371863406
7. Adobe Stock. Text AEFI on white background with various pills and vitamins around, medical concept [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/text-aefi-on-white-background-there-are-various-pills-and-vitamins-around-medical-concept/384835269
8. Adobe Stock. Impfstoff mit Spritze und Impfausweis in einer Arztpraxis [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/impfstoff-mit-spritze-und-impfausweis-in-einer-arztpraxis/499166725
9. Adobe Stock. Hands holding card with text “trusted” representing credibility, integrity, brand loyalty, and quality assurance [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/hands-holding-card-with-text-ltrusted-representing-credibility-integrity-brand-loyalty-and-quality-assurance-message/1382013478
10. Adobe Stock. Medical vaccination line icon continuous line with medicine vaccine sign and pharmacy medication symbol [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/medical-vaccination-line-icon-continuous-line-with-share-plane-medicine-vaccine-sign-pharmacy-medication-symbol-hearts-rate-review-in-speech-bubble-medical-vaccination-single-line-ribbon-vector/1630792550
11. Adobe Stock. Strategizing long-term actions to enhance population health and ensure a healthier future for all [Internet]. Adobe; [cited 2026 May 20]. Available from: https://stock.adobe.com/nl/images/strategizing-long-term-actions-to-enhance-population-health-and-ensure-a-healthier-future-for-all/1627619888
Blog Resources
- 1- kffjoshuam. How Does the Federal Government Monitor Vaccine Safety? | KFF [Internet]. KFF. 2025 [cited 2025 Sep 8]. Available from: https://www.kff.org/other-health/how-does-the-federal-government-monitor-vaccine-safety/
- 2- The Perfect Storm: Measles Resurgence in an Era of Vaccine Disinformation and the Dismantling of Public Health | Milbank Memorial Fund [Internet]. Milbank Memorial Fund. 2025. Available from: https://www.milbank.org/quarterly/opinions/the-perfect-storm-measles-resurgence-in-an-era-of-vaccine-disinformation-and-the-dismantling-of-public-health/
- 3- Prasad V, Makary MA. An Evidence-Based Approach to Covid-19 Vaccination. New England Journal of Medicine. 2025 May 20;
- 4- Pierz AJ, Rauh L, Masoud D, Alanna Kate Cruz, P. Christopher Palmedo, Ratzan SC, et al. Supporting US healthcare providers for successful vaccine communication. Supporting US healthcare providers for successful vaccine communication. 2023 May 2;23(1).
- 5- Communicating About Flu and COVID-19 Vaccination in 2025-26 - Public Health Communications Collaborative [Internet]. Public Health Communications Collaborative. 2025 [cited 2025 Sep 8]. Available from: https://publichealthcollaborative.org/communication-tools/communicating-about-flu-and-covid-19-vaccination-in-2025-26/
- 6- Baden LR, El HM, Essink B, Follmann D, Hachigian G, Strout C, et al. Long-term safety and effectiveness of mRNA-1273 vaccine in adults: COVE trial open-label and booster phases. Nature Communications [Internet]. 2024 Aug 29;15(1). Available from: https://www.nature.com/articles/s41467-024-50376-z
- 7- Xu W, Ren W, Wu T, Wang Q, Luo M, Yi Y, et al. Real-World Safety of COVID-19 mRNA Vaccines: A Systematic Review and Meta-Analysis. Vaccines [Internet]. 2023 Jun 19;11(6):1118–8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10301865/
- 8- Management of Myocarditis and Pericarditis: What Patients Need to Know ESC Clinical Practice Guidelines for the [Internet]. [cited 2025 Sep 8]. Available from: https://www.escardio.org/static-file/Escardio/Guidelines/Documents/ESC-patient-guidelines-Myocarditis-Pericarditis..pdf
- 9- CDC. Grading of Recommendations, Assessment, Development, and Evaluation (GRADE): Updated COVID-19 vaccine (2024-2025 Formulation) [Internet]. Advisory Committee on Immunization Practices (ACIP). 2025 [cited 2025 Sep 8]. Available from: https://www.cdc.gov/acip/grade/covid-19-2024-2025-6-months-and-older.html
- 10- Center. FDA Approves Required Updated Warning in Labeling of mRNA COVID-19 Vac [Internet]. US Food and Drug Administration. 2025. Available from: https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-approves-required-updated-warning-labeling-mrna-covid-19-vaccines-regarding-myocarditis-and
- 11- Meyer S. Update on CDC’s COVID-19 Vaccine Safety Monitoring [Internet]. 2025 [cited 2025 Sep 8]. Available from: https://www.cdc.gov/acip/downloads/slides-2025-06-25-26/04-Meyer-COVID-508.pdf
- 12- McDonald J. Q&A on the 2024-2025 COVID-19 Vaccines - FactCheck.org [Internet]. FactCheck.org. 2024. Available from: https://www.factcheck.org/2024/10/qa-on-the-2024-2025-covid-19-vaccines/
- 13- Wong HL, Hu M, Zhou CK, Lloyd PC, Amend KL, Beachler DC, et al. Risk of myocarditis and pericarditis after the COVID-19 mRNA vaccination in the USA: a cohort study in claims databases. The Lancet [Internet]. 2022 Jun 11;399(10342):2191–9. Available from: https://www.sciencedirect.com/science/article/pii/S0140673622007917
- 14- K. Faksova, Walsh D, Jiang Y, Griffin J, Phillips A, Gentile A, et al. COVID-19 vaccines and adverse events of special interest: A multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine. 2024 Feb 1;42(9).
- 15- Cassard C. 99 Million People Included in Largest Global Vaccine Safety Study - Brighton Collaboration [Internet]. Brighton Collaboration. 2024. Available from: https://brightoncollaboration.org/press-release-gvdn-largest-global-vaccine-safety-study/
- 16- hnesich_drupal. NASEM releases evidence review on COVID-19 vaccine safety | AHA News [Internet]. www.aha.org. Available from: https://www.aha.org/news/headline/2024-04-16-nasem-releases-evidence-review-covid-19-vaccine-safety
- 17- Soe P, Vanderkooi OG, Sadarangani M, Naus M, Muller MP, Kellner JD, et al. mRNA COVID-19 vaccine safety among children and adolescents: a Canadian National Vaccine Safety Network cohort study. The Lancet Regional Health - Americas. 2024 Nov 27;40:100949–9.
- 18- Olawade DB, Teke J, Oluwaseun Fapohunda, Weerasinghe K, Usman SO, Ige AO, et al. Leveraging artificial intelligence in vaccine development: A narrative review. Journal of Microbiological Methods. 2024 Sep 1;224:106998–8.
- 19- Research C for BE and. COVID-19 Vaccine Safety Surveillance. Center for Biologics Evaluation and Research [Internet]. 2021 Feb 9; Available from: https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/covid-19-vaccine-safety-surveillance
- 20- IntuitionLabs. AI Applications in Pharmacovigilance and Drug Safety [Internet]. IntuitionLabs. 2025. Available from: https://intuitionlabs.ai/articles/ai-pharmacovigilance-drug-safety
- 21- Cherkas Y, Ide J, van Stekelenborg J. Leveraging Machine Learning to Facilitate Individual Case Causality Assessment of Adverse Drug Reactions. Drug Safety. 2022 May;45(5):571–82.
- 22- Area Under Curve (AUC) [Internet]. Iterate.ai. 2025 [cited 2025 September 9]. Available from: https://www.iterate.ai/ai-glossary/what-is-auc-metric-explained
- 23- Understanding Area Under the Curve (AUC) in Model Performance [Internet]. Lyzr. 2024. Available from: https://www.lyzr.ai/glossaries/area-under-the-curve/
- 24- Tools and innovations [Internet]. www.who.int. Available from: https://www.who.int/teams/regulation-prequalification/regulation-and-safety/pharmacovigilance/guidance/operations/tools-innovations
- 25- Early warning system [Internet]. www.who.int. Available from: https://www.who.int/groups/global-advisory-committee-on-vaccine-safety/topics/covid-19-vaccines/early-warning-system
- 26- Blau EF. Progress in Immunization Safety Monitoring — Worldwide, 2020–2022. MMWR Morbidity and Mortality Weekly Report [Internet]. 2023;72. Available from: https://www.cdc.gov/mmwr/volumes/72/wr/mm7249a2.htm
- 27- Research C for BE and. COVID-19 Vaccine Safety Surveillance. Center for Biologics Evaluation and Research [Internet]. 2021 Feb 9; Available from: https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/covid-19-vaccine-safety-surveillance
- 28- ICMRA Vaccine Safety Collaboration Workshop | International Coalition of Medicines Regulatory Authorities (ICMRA) [Internet]. Icmra.info. 2017 [cited 2025 September 10]. Available from: https://www.icmra.info/drupal/covid-19/13january2021
- 29- Kohl KS, Bonhoeffer J, Braun MM, Chen RT, Duclos P, Heijbel H, et al. The Brighton Collaboration: Creating a Global Standard for Case Definitions (and Guidelines) for Adverse Events Following Immunization [Internet]. Henriksen K, Battles JB, Marks ES, Lewin DI, editors. PubMed. Rockville (MD): Agency for Healthcare Research and Quality (US); 2005. Available from: https://www.ncbi.nlm.nih.gov/books/NBK20507/
- 30- Deady M, Duncan R, Jones LD, Sang A, Goodness B, Pandey A, et al. Data quality and timeliness analysis for post-vaccination adverse event cases reported through healthcare data exchange to FDA BEST pilot platform. Frontiers in public health. 2024 July 8;12.
- 31- kfflizw. Kindergarten Routine Vaccination Rates Continue to Decline | KFF [Internet]. KFF. 2025. Available from: https://www.kff.org/medicaid/kindergarten-routine-vaccination-rates-continue-to-decline/
- 32- Centers for Disease Control and Prevention. Measles cases and outbreaks [Internet]. Measles (Rubeola). CDC; 2025. Available from: https://www.cdc.gov/measles/data-research/index.html
- 33- Ilyasova AA, Queen TL, Gilkey M, Fogel BN, Odebunmi OO, Yanguela J, et al. Use of presumptive recommendations and other strategies to encourage HPV vaccine uptake: Results from a national survey of primary care health professionals. PLoS ONE [Internet]. 2025 Aug 4 [cited 2025 Sep 10];20(8):e0327872–2. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12321062/
- 34- Andersson, N.W., Svalgaard, I.B., Hoffmann, S.S., & Hviid, A. (2025). Aluminum-Adsorbed Vaccines and Chronic Diseases in Childhood: A Nationwide Cohort Study. Annals of Internal Medicine. Aluminum-Adsorbed Vaccines and Chronic Diseases in Childhood: Annals of Internal Medicine: Vol 0, No 0.
- 35- Cima.care. 2025 [cited 2025 Sep 10]. Available from: https://www.cima.care/post?id=43
- 36- Cima.care. 2025. Available from: https://www.cima.care/post?id=40
- 37- Cima.care. 2025 [cited 2025 Sep 10]. Available from: https://www.cima.care/post?id=34
- 38- Cima.care. 2025 [cited 2025 Sep 10]. Available from: https://www.cima.care/post?id=42
- 39- Tagbo BN, Ejekam CS, Oppong-Amoako W, Yameogo TM, Mitiku A, Esangbedo DO, et al. Strengthening Vaccine Safety Systems, Research, and Regional Collaboration in Africa: A Call to Action. Vaccines [Internet]. 2025 Jun 19 [cited 2025 Jul 18];13(6):661. Available from: https://www.mdpi.com/2076-393X/13/6/661
- 40- K. Faksova, Walsh D, Jiang Y, Griffin J, Phillips A, Gentile A, et al. COVID-19 vaccines and adverse events of special interest: A multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine. 2024 Feb 1;42(9).
- 41- Team DigitalDefynd. Role of AI in Vaccine Development and Distribution [2025] [Internet]. DigitalDefynd. 2025 [cited 2025 Sep 10]. Available from: https://digitaldefynd.com/IQ/ai-in-vaccine-development-distribution/
Enjoyed this article?
Share it with your friends on LinkedIn: Vaccine Safety: Evidence, Controversies, and Communication
Follow us on LinkedIn for more updates and insights: Cima Care GmbH
