Balancing Safety with Critical Protection in Genetically Vulnerable Children |
August 26, 2025 . 5 Minutes read
Safe and Essential Vaccination for Children with Genetic Disorders
Vaccination for children with genetic disorders represents one of the most critical yet misunderstood areas in pediatric medicine, where evidence-based protection meets complex clinical decision-making. Recent research shows that personalized vaccination strategies are not only safe but essential for children with genetic conditions, providing robust protection while maintaining outstanding safety profiles when proper protocols are followed. Current evidence demonstrates that the primary challenge is no longer determining whether to vaccinate these vulnerable children, but rather how to optimize individualized approaches that balance their enhanced infection risks with their unique immune responses, transforming fear-based hesitation into confidence-driven protection.
General principles across genetic disorders
Vaccination approaches for children with genetic disorders follow fundamental principles that guide clinical decision-making. While evidence supports vaccination across all genetic conditions, implementation challenges persist in translating guidelines into practice.
- Standard vaccination schedules remain the foundation for most children with genetic disorders. The universal principle is that these conditions themselves do not contraindicate vaccination; rather, specific complications or treatments may require modifications. Live vaccines represent the primary consideration, particularly for conditions involving immunocompromise or immunosuppressive treatments. [14]
- Risk-benefit analysis consistently favors vaccination in these populations. Children with genetic disorders often face higher baseline risks from infections due to organ system involvement, immune dysfunction, or treatment effects. Multiple studies demonstrate that vaccine-preventable diseases pose greater threats than vaccination risks in these vulnerable populations. [15]
- Clinical practice gaps persist despite clear guidelines. Major health authorities, including the Centers for Disease Control and Prevention (CDC), WHO, and American Academy of Pediatrics (AAP), have established condition-specific protocols that contraindicate live vaccines only in severe immunodeficiencies, whether induced by genetic disorders or not, while strongly recommending enhanced protection through additional doses of inactivated vaccines. [1, 2 & 3] However, real-world vaccination rates in children with genetic disorders remain concerning and significantly lower compared to healthy children, [4] highlighting the critical gaps between evidence-based recommendations and clinical practice and the importance of targeted education for both parents and pediatricians to improve vaccination uptake in children with genetic disorders.
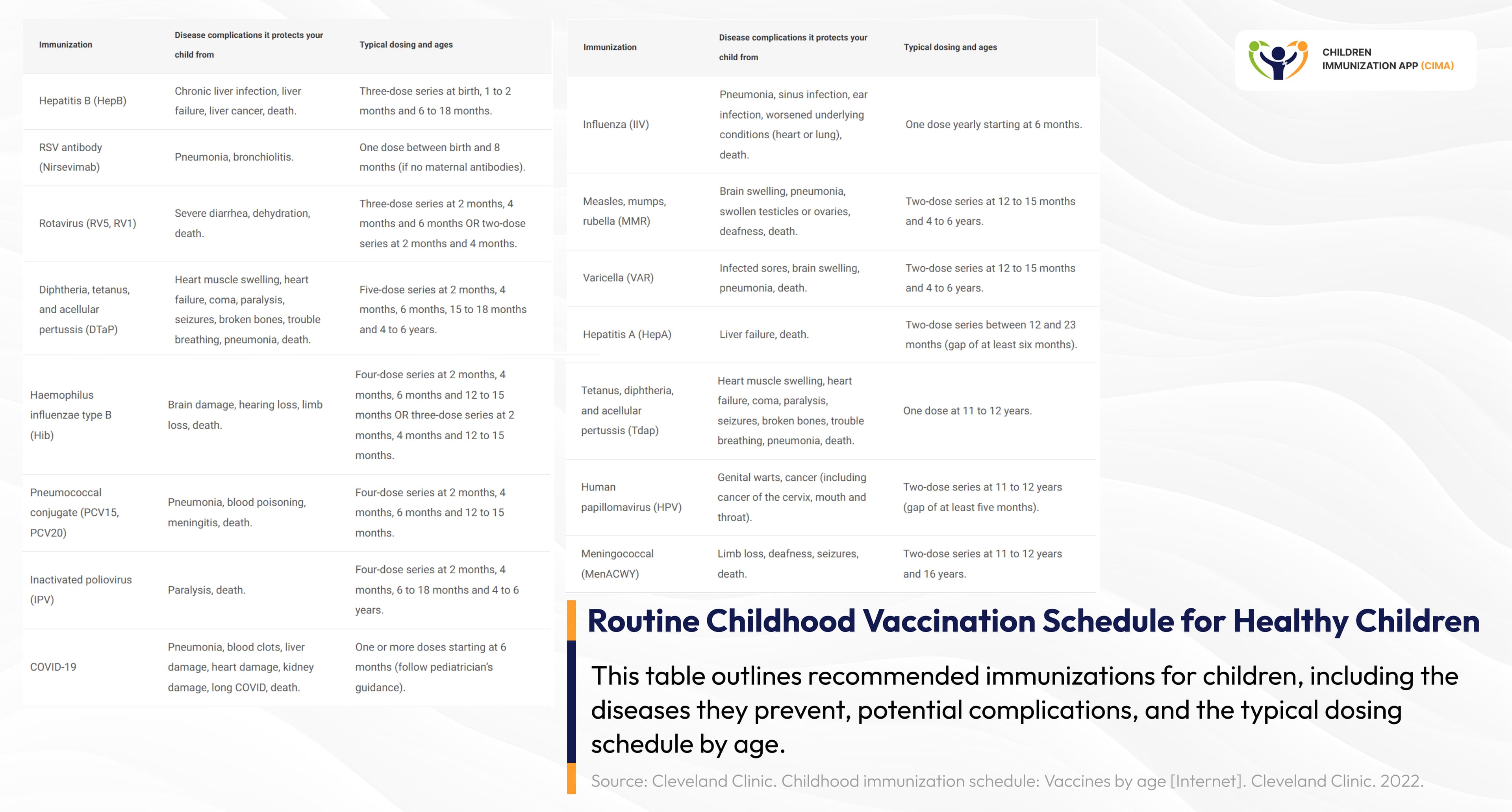
Schedule of standard childhood vaccinations outlining routine immunizations and timing by age.
Metabolic Disorders Demonstrate Vaccine Safety Profiles
- Metabolic disorder patients show normal vaccine responses with standard schedules, contradicting historical concerns about triggering disease exacerbations. [7]
- A 2023 study in the Journal of Paediatrics and Child Health found no cases of acute metabolic decompensation after COVID‑19 vaccination in children with inborn errors of metabolism, including phenylketonuria (PKU). The only observed effect was a brief rise in phenylalanine levels in two PKU patients (of 36 vaccination episodes in 18 patients) 24 hours after the second vaccine dose of COVID-19 vaccines, without any clinical complications, supporting the vaccine's safety in this group. [8]
- Research on metabolic disorders provides compelling evidence for vaccination safety. Urea cycle disorders (UCDs) are genetic disorders that cause deficiencies in one or more of the enzymes required for the urea cycle. Patients with UCDs are subject to hyperammonemic episodes (HAEs) after infection, fever, or other stressors. Studies found no association between vaccination and hyperammonemic episodes, with the Safety Assessment Network establishing the safety foundation for this medically vulnerable population. [16]
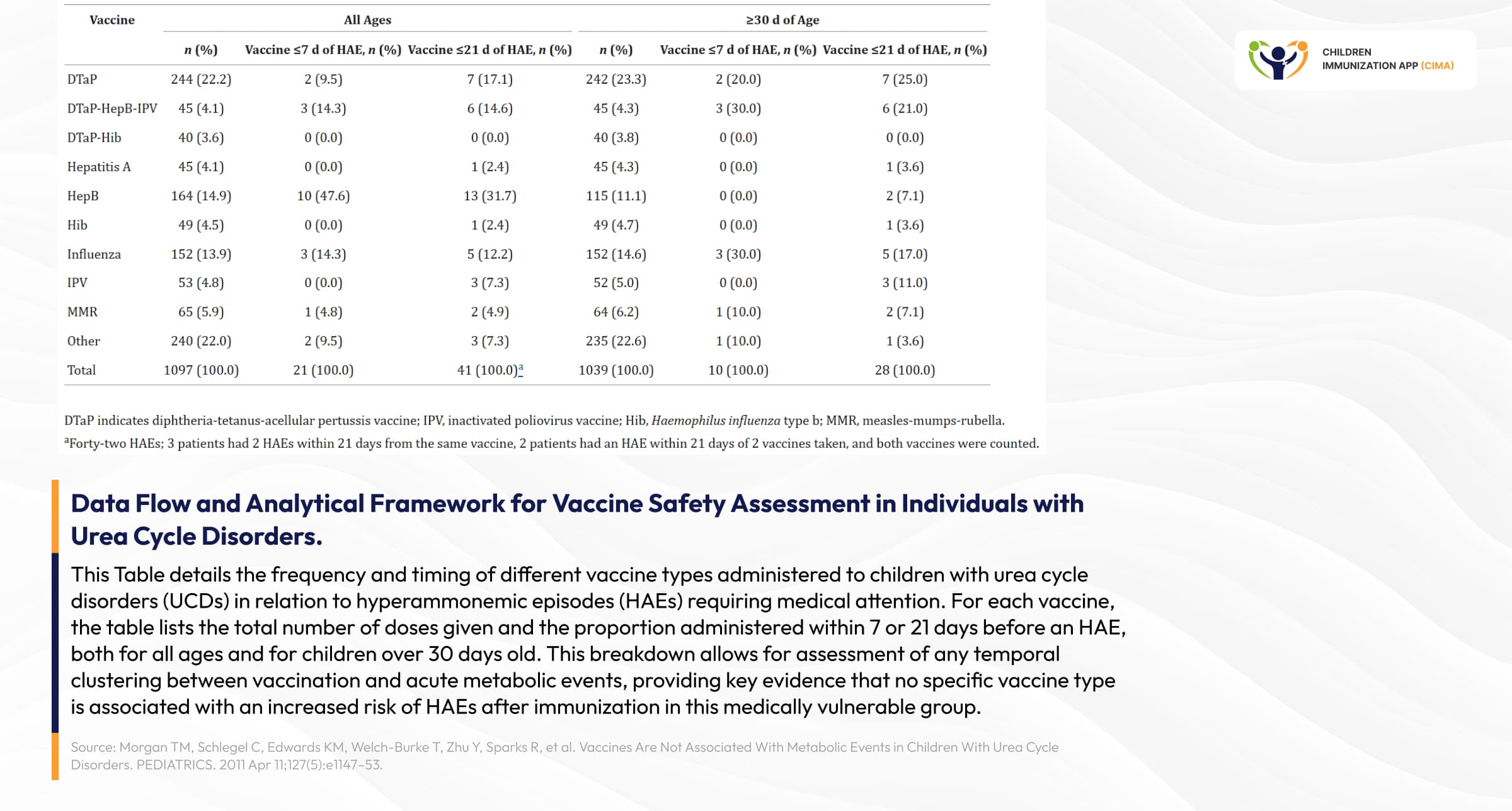
Research data showing vaccine timing vs metabolic episodes in urea cycle disorders, assessing any temporal clustering between vaccination and acute metabolic events
Standard Vaccination Protocols for Chromosomal Disorders: From Down Syndrome to Turner, Edwards, and Patau Syndromes
- Down syndrome represents the most studied chromosomal disorder for vaccination purposes. American Academy of Pediatrics (AAP) guidelines (2022) recommend standard immunization schedules plus additional vaccines: annual influenza vaccination and 23-valent pneumococcal polysaccharide vaccine for children with chronic cardiac or pulmonary disease. The key finding is the fact that children with Down syndrome are NOT classified as immunocompromised for vaccination purposes, making live vaccines generally safe, unless there are specific contraindications. [17]
- The research reveals important distinctions in immune responses across genetic conditions. Individuals with Down syndrome show a 4-5 times higher COVID-19 hospitalization risk and are 10x more likely to die due to COVID-19, but maintain robust vaccine responses, supporting prioritized vaccination schedules. In 2024, a study confirmed that COVID-19 vaccination in this group is safe and effective, with over half reporting no side effects after the first and second doses. The most common mild reactions were injection site pain, fatigue, and fever, all occurring at low rates. [9, 10 & 11]
- Coverage disparities present significant challenges for children with Down syndrome. According to a 2020 study (Parent Attitudes about Childhood Vaccines Survey, PACV, was used to assess vaccine hesitancy among parents of children with Down syndrome), only 58% of children with Down syndrome were up to date for the full combined 7-vaccine series by 19 months of age. This suboptimal coverage was strongly linked to vaccine hesitancy among parents, who reported concerns about vaccine safety and potential effects on their child's underlying health. The study emphasized that these gaps highlight a critical opportunity for targeted educational strategies and communication interventions aimed at improving immunization rates in this vulnerable population, ensuring better protection against preventable diseases. [18]
- Children with Turner syndrome (occurring in females who are missing all or part of one X chromosome, affecting about 50 per 100,000 females, and requiring lifelong, multidisciplinary care due to its impact on multiple organs) should generally follow the standard pediatric immunization schedule. There are no Turner-specific vaccine contraindications, but careful cardiac assessment is advised for those with congenital heart disease. [19]
- In children with trisomy 18 (Edwards syndrome, with severe developmental delays and heart defects) and trisomy 13 (Patau syndrome, with brain abnormalities and facial malformations), routine immunization schedules can be followed when clinically appropriate, but decisions about vaccination should be individualized, considering the child's medical stability and prognosis. [20]

Vaccination guidance flowchart for a few major chromosomal disorders and safety considerations.
Neuromuscular Disorders: Understanding Steroid-Related Vaccination Considerations
- Corticosteroid therapy creates the primary vaccination consideration in neuromuscular disorders due to its immunosuppressive effects. Many children with conditions like Duchenne muscular dystrophy (caused by mutations in the DMD gene on the X chromosome, affecting dystrophin production), inflammatory myopathies(an autoimmune condition where the body’s immune system attacks muscle tissue), and certain forms of spinal muscular atrophy (caused by mutations in the SMN1 gene) require chronic corticosteroid treatment to slow disease progression, reduce inflammation, or manage symptoms. However, these life-saving medications significantly suppress the immune system, creating important implications for vaccine safety and timing. [21]
- Understanding steroid-induced immunosuppression is crucial for vaccination decisions. Corticosteroids suppress both cellular and humoral immunity by reducing T-cell function, decreasing antibody production, and impairing the body's ability to mount effective immune responses. This immunosuppression makes patients more susceptible to infections while simultaneously creating risks from live vaccines, which contain weakened but living pathogens that could potentially cause disease in immunocompromised individuals. [22]
- The CDC and infectious disease societies have precisely defined high-dose steroid immunosuppression criteria. In children, immunosuppressive doses include ≥2 mg/kg/day prednisone or ≥2.4 mg/kg/day deflazacort (the preferred steroid for Duchenne muscular dystrophy due to better bone health profiles). For adolescents and adults, the threshold is ≥20 mg/day prednisone or ≥24 mg/day deflazacort. These doses represent levels that significantly compromise immune function and require modified vaccination approaches. [23]
- Live vaccines are contraindicated during high-dose daily steroids because they contain attenuated (weakened) viruses or bacteria that can potentially cause infection in immunocompromised patients. Live vaccines include MMR (measles, mumps, and rubella), varicella (chickenpox), live influenza (FluMist), rotavirus, and yellow fever vaccines. In contrast, inactivated vaccines (containing killed pathogens) and subunit vaccines remain safe and are strongly recommended, including COVID-19 vaccines, inactivated influenza, Tdap, and pneumococcal vaccines. [12]
- Safe vaccination timing requires careful steroid management. Live vaccines are permitted when steroid doses are reduced below immunosuppressive levels for at least one month before vaccination, and patients should maintain these lower doses for one month after vaccination to ensure adequate immune response. This timing allows the immune system to partially recover while maintaining vaccine effectiveness. For urgent vaccination needs, inactivated vaccines can be administered at any time during steroid therapy, though immune responses may be reduced. [29]

Clinical guide categorizing corticosteroid immunosuppression levels, helping assess vaccination timing.
Vaccine Safety and Efficacy in Immunocompromised Children
Altered immunocompetence refers to conditions, whether congenital, acquired, or drug-induced, that affect immune system function, increasing risk from infections and live vaccines.[12]
General Principles
- Risks: Live vaccines can cause uncontrolled replication in significantly immunocompromised patients. Inactivated vaccines are generally safe but may have reduced efficacy.
- Guidance: Decisions should be individualized, and clinicians should consult with relevant specialists when uncertain.
Conditions Associated with Altered Immunocompetence
- Primary immunodeficiencies: Including severe combined immunodeficiency (SCID), X-linked agammaglobulinemia, DiGeorge syndrome, and complement deficiencies.
- Secondary or acquired immunodeficiency: Due to HIV infection, cancer chemotherapy, hematopoietic stem cell transplant, radiation, or immunosuppressive therapy (including high-dose corticosteroids).
- Functional or anatomic asplenia: Includes sickle cell disease, congenital asplenia, or splenectomy, with increased risk for certain encapsulated bacterial infections.
Vaccination of Persons with Altered Immunocompetence
- Live vaccines: Contraindicated in significantly immunocompromised individuals; may be considered in those with less severe compromise after specialist consultation.
- Inactivated vaccines: Safe and recommended, though immune responses may be suboptimal. Revaccination may be required after immune recovery (e.g., after chemotherapy or transplant).
- Household contacts: Should be fully vaccinated to protect immunocompromised persons, and live vaccines for contacts are generally safe (with specific precautions noted for some vaccines). [12]
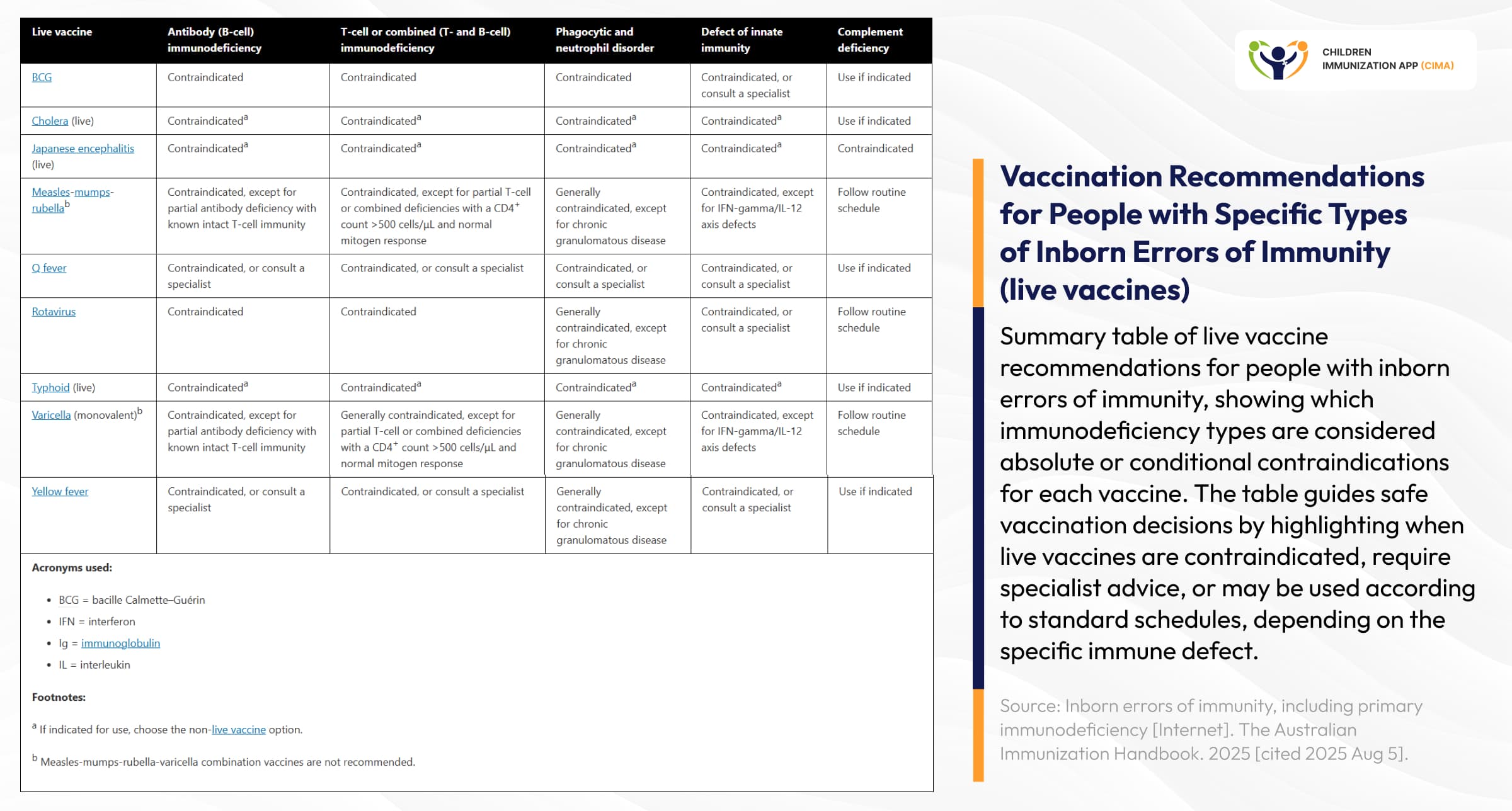
Clinical reference matrix for live vaccine safety across different immunodeficiency types.
Vaccines Contraindicated in Immunocompromised Persons
MMR, varicella, LAIV (live attenuated influenza vaccine), yellow fever, oral polio (no longer used in the US), and live-attenuated zoster vaccine (Zostavax) are contraindicated in significantly immunocompromised persons.
Vaccination of Specific Patient Groups
- HIV-infected children and adults: Inactivated vaccines are safe and recommended; some live vaccines (e.g., MMR, varicella) may be considered if immune status is adequate (CD4 counts/percentages above thresholds).
- Cancer patients and transplant recipients: Vaccines are ideally administered before therapy or transplantation. Inactivated vaccines are only given after therapy, when immune function recovers; live vaccines are generally avoided or postponed.
- Patients on immunosuppressive therapy: Timing of vaccination and immunosuppressive regimens requires close coordination—live vaccines are generally avoided, and inactivated vaccines may be less effective.
Healthcare Personnel and Household Contacts
- Household members and healthcare workers may receive live vaccines unless severely immunosuppressed, to reduce disease transmission risks to vulnerable patients.
Additional Guidance
- Documentation and communication: Clearly record immunization decisions and rationale; consult immunization experts or guidelines when doubt exists.
- Specialist involvement: Immunization plans for complex patients should be developed collaboratively with appropriate subspecialists. [12 & 13]
COVID-19 Vaccination Series
Vaccine safety studies involving immunocompromised children report only mild, transient side effects and no elevated risk of serious adverse events compared to healthy children. [5] 2025 studies confirm that immunocompromised children benefit significantly from additional COVID-19 vaccine doses. The evidence shows that while their immune response to the initial vaccination series may be weaker compared to healthy children, administering extra primary or booster doses leads to a marked increase in both antibody and cellular immune responses. This enhanced immunogenicity after additional doses helps bring their level of protection much closer to that observed in non-immunocompromised peers, providing critical reassurance for this vulnerable group. [6]

Evidence-based vaccination protocols categorized by specific immunodeficiency types.
Neurogenetic disorders that require individualized assessment
Neurogenetic disorders that require personalized evaluation include:
- Autism spectrum disorders (ASD) with a genetic basis show no increased vaccination risk. The 2023 study of 1,335 children (762 with neurodevelopmental disorders) who received the SARS-CoV-2 vaccination found no specific neurodevelopmental (ND) diagnosis that increased the risk of adverse effects.
- Attention-Deficit/Hyperactivity Disorder (ADHD) and ASD were NOT associated with adverse outcomes, with researchers concluding that "children with ND can be reassured that the SARS-CoV-2 vaccination is a safe regimen. [24]
- Genetic epilepsy syndromes, such as Dravet syndrome, require tailored vaccination approaches to ensure both safety and optimal disease management. Dravet syndrome, a severe genetic epilepsy typically linked to variants in the SCN1A gene, carries the highest risk of vaccine-associated seizures among childhood epilepsies: approximately one-third of children with Dravet syndrome experience their first seizure following routine vaccination, often in infancy. However, current evidence, including Australian and international data, consistently shows that the long-term outcomes for these children do not differ based on whether the first seizure is temporally linked to vaccination or not. Vaccination is still broadly recommended for children with Dravet syndrome because the benefits of immunization, in preventing infections that themselves frequently trigger seizures and can worsen neurologic outcomes, far outweigh the risks. [25 & 26]
Hospital-Based Vaccination Protocol for Children with Dravet Syndrome
- Children with Dravet syndrome or severe vaccine-proximate seizures should be vaccinated following a specialist protocol in a hospital immunization clinic or as a closely monitored day admission.
- Emergency seizure management medications (such as midazolam or rectal diazepam) should be readily available, and individualized rescue plans implemented as per each child's epilepsy action plan.
- Routine pre-vaccination Prophylactic antipyretics or antiepileptic medications are not recommended for all cases; decisions should be individualized.
- A multidisciplinary team, combining expertise from immunization specialists and the child's neurologist, should coordinate care, monitor and manage any adverse events, and ensure prompt intervention if needed.
- Parents and carers should be reassured that vaccination-triggered seizures do not result in worse long-term seizure control or developmental outcomes.
- With these safeguards, vaccination is not contraindicated in Dravet syndrome, and standard immunization schedules can usually be followed safely. [25 & 26]
Recent research confirms that children with Fragile X syndrome (FXS) should follow standard pediatric vaccination protocols, as there are no specific contraindications to any routine or COVID-19 vaccines for this population. FXS is caused by mutations in the FMR1 gene, resulting in a lack of FMRP protein and characterized by impaired brain development and functional abnormalities. Studies show that children and young adults with FXS are as likely, or in some cases even more likely, to be fully vaccinated as their peers, although disparities exist across demographic groups. Immune system alterations have been observed in FXS, including increased susceptibility to infections and reduced immune responsiveness. Still, these factors have not been shown to increase vaccine risk or require schedule modifications. The safest and most reliable approach remains adherence to established immunization guidelines, ensuring protection against preventable diseases. There is no research or expert consensus indicating increased adverse reactions to vaccination in FXS COVID-19 vaccines are also advised for these children without restriction. [27 - 31]
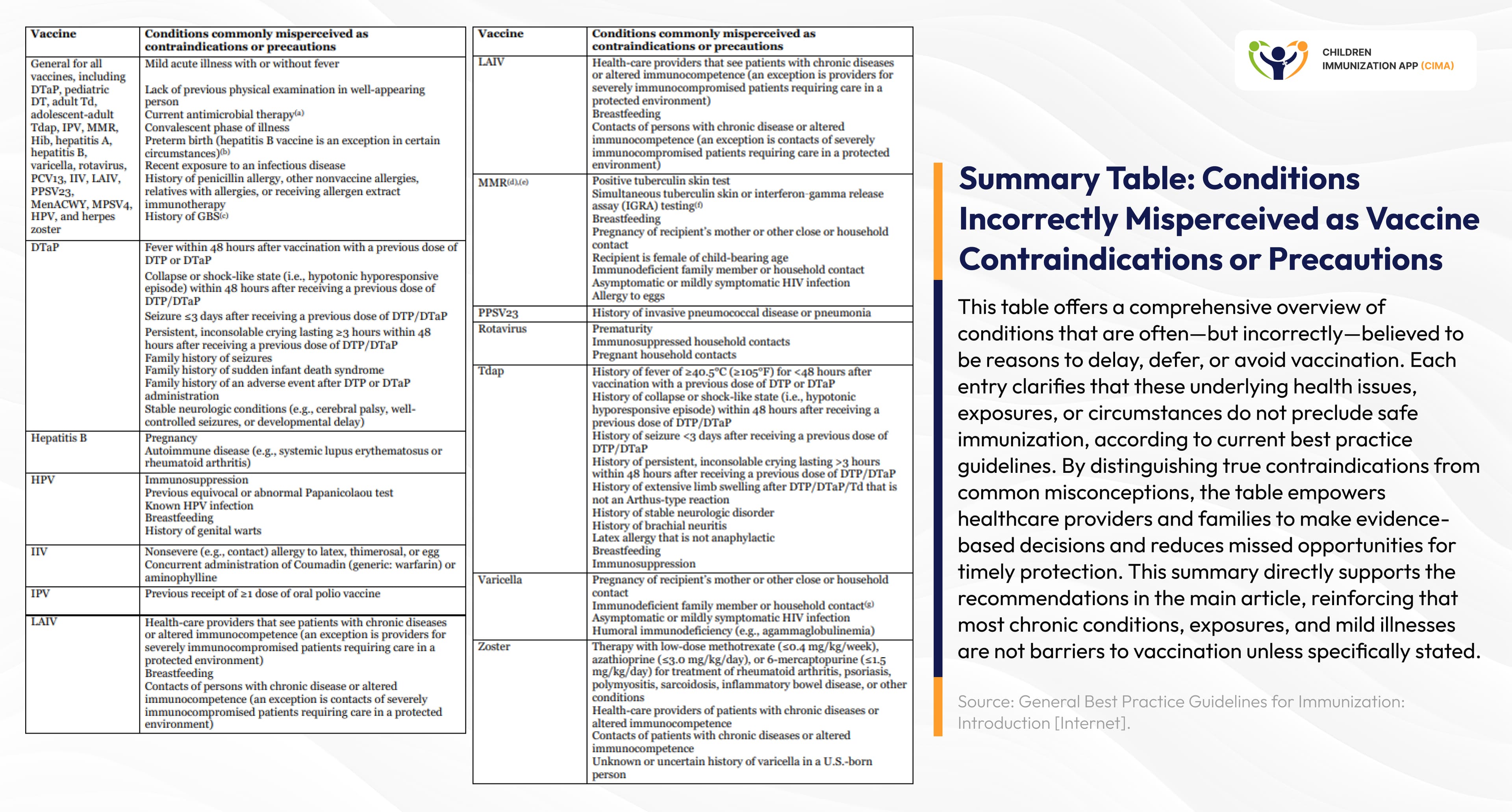
Clinical table clarifying conditions incorrectly viewed as vaccine barriers.
Vaccination in Connective Tissue Disorders: Safety and Best Practice
- Ehlers-Danlos syndrome (EDS) is a group of inherited connective tissue disorders caused by genetic defects affecting collagen, leading to symptoms such as hypermobile joints, fragile skin, easy bruising, and poor wound healing. Vaccination, including for COVID-19, is generally considered safe for people with EDS and hypermobility spectrum disorders (HSD). Recent advice from the Ehlers-Danlos Society highlights that, for most individuals with these conditions, the protective benefits of immunization outweigh potential risks related to tissue fragility. [32] A 2024 survey showed that among 368 people with hypermobile EDS, 87.2% experienced mild, expected vaccine reactions, and only 3.1% required emergency care. [33]
- Marfan syndrome is another genetic connective tissue disorder caused by mutations in the fibrillin-1 (FBN1) gene. This mutation leads to abnormal connective tissue throughout the body, affecting the skeleton, eyes, heart, blood vessels, and more. Children and adults with Marfan syndrome and osteogenesis imperfecta (OI) should receive all recommended vaccines according to the standard immunization schedule, as there are no condition-specific vaccine contraindications for either disorder. However, special attention should be paid to cardiovascular risks in Marfan syndrome and to bone fragility in OI, meaning vaccinations should be administered with gentle handling and, when necessary, tailored positioning. Clinical reviews and specialist groups confirm that careful monitoring during and after vaccination is important, but immunization is safe and effective for children with Marfan syndrome and OI. [35 & 36]

Essential pre-vaccination screening checklist ensuring patient safety and readiness.
Genetic Syndromes Span the Immunocompetence Spectrum
- Williams syndrome (WS) is caused by a microdeletion on chromosome 7q11.23, which includes the elastin (ELN) gene, contributing to many of the vascular and connective tissue features of the syndrome, such as soft, lax skin; joint hypermobility in early life (which may become stiffness later); and vascular issues like supravalvular aortic stenosis (SVAS). Additionally, WS causes symptoms like a distinctive facial appearance and developmental delays. Health agencies have not issued any special immunization contraindications or schedule changes solely due to Williams syndrome. In practice, children with Williams syndrome are expected to receive vaccines on the routine childhood schedule unless other individual medical issues warrant a change. Notably, the Williams Syndrome Foundation (UK) notes that “childhood vaccinations are just as important for your child as any other child” and that a Williams syndrome diagnosis “should have no impact on them receiving these vaccinations.” This reflects the absence of any syndrome-specific vaccine guidelines, implying normal immunization timing and precautions should be followed. [37] The Williams Syndrome Association advises that individuals aged 5 and up with WS should get the COVID-19 vaccine. WS itself is not a contraindication, though rare unrelated issues (like severe allergies or immunosuppression) may require individual medical guidance. [41] Moreover, limited research suggests that children with Williams syndrome mount normal immune responses to vaccines. In a small clinical study evaluating the H1N1 influenza vaccine, children and adolescents with WS showed immune protection and tolerated the vaccine as well as typically developing children. [38]
- Prader-Willi syndrome (PWS) is caused by the loss of function of genes on the paternal copy of chromosome 15q11–q13, often due to deletion or uniparental disomy. These children show mild to moderate intellectual disability, hypotonia (low muscle tone) at birth, and other symptoms. Children with Prader-Willi (PWS) generally follow the standard childhood vaccination schedule. Medical guidelines emphasize that routine vaccines should be administered to PWS patients just as in other children, unless standard contraindications exist. [39] Infants with PWS have severe hypotonia (low muscle tone), weak swallow, and poor cough, putting them at higher risk of serious respiratory infections. Given this vulnerability, preventing respiratory illnesses is critical. Routine childhood vaccines like Prevnar-13 (pneumococcal conjugate) and Hib (Haemophilus influenzae type b), which protect against common bacterial pneumonias, are especially beneficial and are part of standard pediatric care. [40 & 42] COVID-19 vaccines (which in most pediatric cases are mRNA-based) are also advised for people with PWS according to expert guidance. Notably, the Prader-Willi Syndrome Association UK states that children with PWS are “not at increased risk from COVID-19 unless they have other medical conditions.” [43]
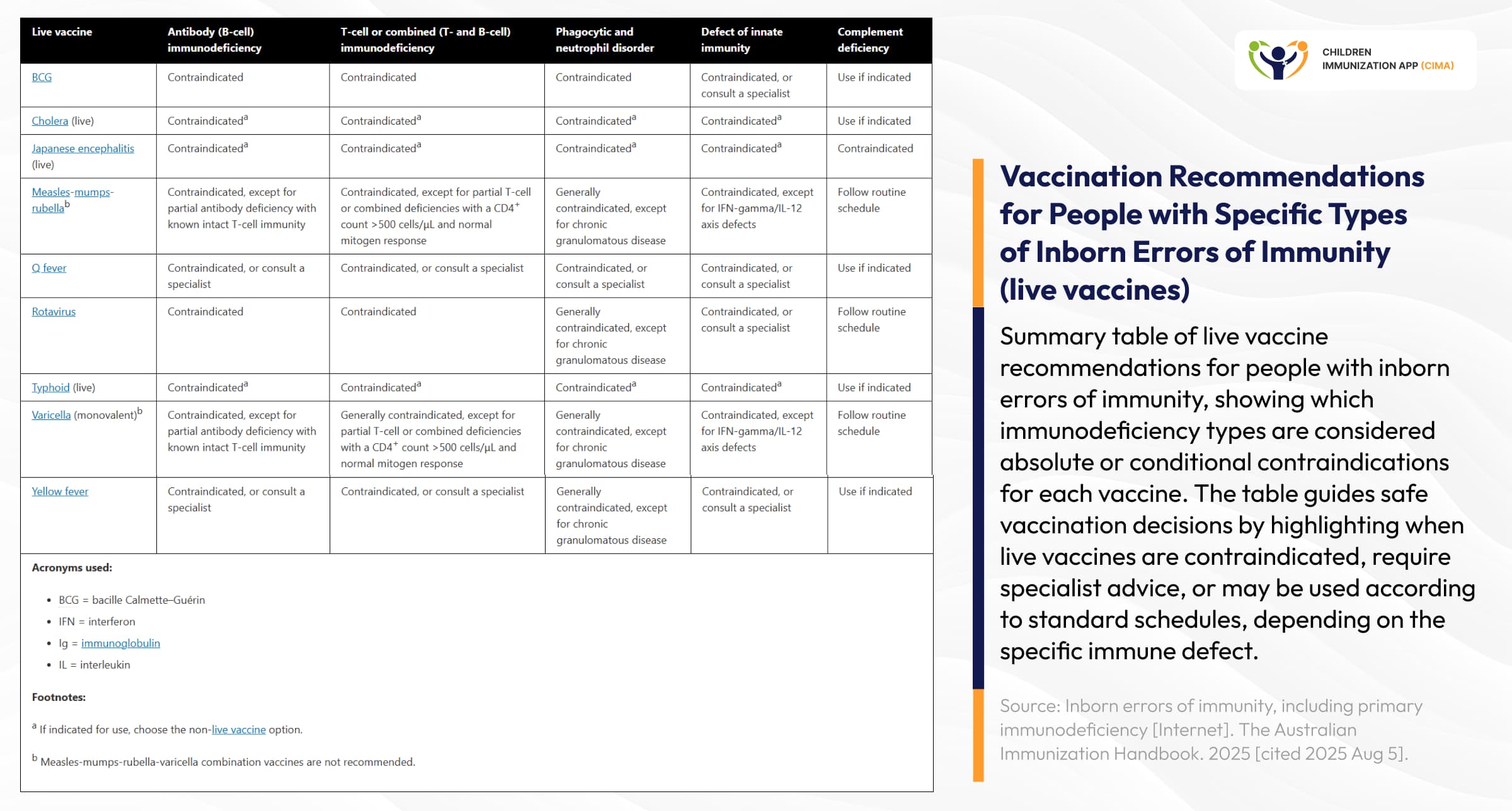
Systematic guide differentiating absolute vaccine contraindications from clinical precautions.
Clinical Practice Recommendations: A Concise Briefing
- Pre-vaccination assessment should include a complete medical history review, assessment of current medications (particularly immunosuppressants), immune status evaluation when indicated, and coordination with subspecialty teams. Universal principles include following standard CDC/AAP schedules unless specific contraindications exist, providing additional vaccines based on underlying conditions, and addressing family concerns with evidence-based information. [44, 45 & 46]
- Special populations require modified approaches. Children with some chromosomal deletions need immune evaluation before live vaccines. [47] Genetic epilepsy syndromes may require specialist immunization clinics. Steroid-treated patients need careful timing of live vaccines. [48] Documentation and surveillance remain crucial for monitoring safety and effectiveness. [49]
- Family education and counselling represent critical interventions. Healthcare providers must address vaccine hesitancy with authoritative evidence while acknowledging legitimate concerns about children with complex medical conditions. [50] The scientific consensus overwhelmingly supports vaccination while recognizing the need for individualized approaches. [51]

Comprehensive clinical framework ensuring safe vaccination for children with genetic disorders.
United Evidence: Comprehensive Vaccination Benefits Across All Genetic Disorders
The evidence unequivocally demonstrates that vaccination represents a critical protective intervention for children with genetic disorders, with careful attention to condition-specific considerations, ensuring optimal safety and effectiveness. Healthcare providers should maintain strong vaccination recommendations while implementing individualized approaches that address the unique needs of this vulnerable population. [55 & 56]

Evidence-based care protects children with genetic conditions through safe vaccination.

Future vision: personalized vaccination strategies integrating genetic profiling and big data.
Research priorities and future directions
- Significant research gaps persist across multiple domains. Long-term vaccine effectiveness data are needed for specific genetic disorders, optimal dosing schedules require investigation, and novel vaccine approaches for immunocompromised subgroups warrant development. [52]
- Emerging considerations include mRNA vaccine technology applications, personalized vaccination strategies based on genetic profiling, and integration with precision medicine approaches. [53]
- Global health equity initiatives must address access disparities for children with genetic disorders worldwide. [54]
CIMA Care: Empowering Specialized Care through Digital Innovation
The complex vaccination needs of children with genetic disorders demand the kind of comprehensive, technology-driven support that CIMA Care provides. Our integrated digital ecosystem addresses the critical gaps identified in current research, ensuring that genetically vulnerable children receive the individualized care they require.
- CIMA App's advanced tracking capabilities become essential for children with genetic disorders who require modified vaccination schedules, enhanced monitoring protocols, and careful coordination between genetic specialists and primary care providers. Our automated reminder system ensures no critical vaccines are missed, while our defaulter identification feature provides the systematic follow-up that research shows is crucial for achieving coverage parity with populations.
- CIMA Health Academy's CPD-certified courses directly address the knowledge gaps that contribute to vaccination hesitancy among healthcare providers caring for children with genetic conditions. Our evidence-based training modules on vaccination science, implementation strategies, and special population considerations empower healthcare professionals with the specialized expertise needed to make informed risk-benefit assessments and communicate effectively with concerned families. in these populations. Children with genetic disorders often face higher baseline risks from infections due to organ system involvement, immune dysfunction, or treatment effects. Multiple studies demonstrate that vaccine-preventable diseases pose greater threats than vaccination risks in these vulnerable populations. [15]
- The data visualization and analytics tools within our platform enable enhanced monitoring protocols that experts universally recommend for genetically vulnerable children. Healthcare providers can track vaccination responses, monitor for adverse events, and generate the detailed documentation required for complex genetic cases, all while contributing to the growing evidence-based knowledge that informs future care standards.
- Through multilingual SMS education, CIMA Care addresses the communication challenges that research identifies as critical barriers to vaccination acceptance. Our WHO-validated content helps families understand why vaccination remains essential despite genetic vulnerabilities, countering misinformation while building the trust necessary for informed decision-making.
As the evidence overwhelmingly supports vaccination for children with genetic disorders while acknowledging the need for individualized approaches, CIMA Care stands ready to bridge the gap between complex clinical guidelines and practical implementation.
Join CIMA Care in transforming specialized pediatric vaccination delivery. Visit www.cima.care to discover how our comprehensive platform supports the complex care needs of children.
Together, we can ensure that every genetically vulnerable child receives the protection they need through safe, evidence-based vaccination strategies.

Integrated platform supporting vaccination needs for all children, including complex cases.
Image References
- 1- Adobe Stock. Vaccine selection. Ampoules with Covid‑19 vaccine in laboratory. Fight with the coronavirus pandemic [Internet]. Adobe Stock; 2021 [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/vaccine-selection-ampoules-with-covid-19-vaccine-in-laboratory-fight-with-the-coronavirus-pandemic/410014447
- 2-ARMMY PICCA. Concept fight against virus COVID-19, doctor or scientist in laboratory holding a syringe with liquid vaccines [Internet]. Adobe Stock; 2021 [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/concept-fight-against-virus-covid-19-corona-virus-doctor-or-scientist-in-laboratory-holding-a-syringe-with-liquid-vaccines-for-children-or-older-adults-concept-diseases-medical-care-science/475900472
- 3- Adobe Stock. Gene therapy technology: current innovations, translational challenges and future perspectives in targeted genetic intervention for monogenic disorders, complex diseases [Internet]. Adobe Stock; 2023 [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/gene-therapy-technology-current-innovations-translational-challenges-and-future-perspectives-in-targeted-genetic-intervention-for-monogenic-disorders-complex-diseases/1555428539
- 4- Alernon77. Medical hands holds syringe and vaccine [Internet]. Adobe Stock; 2021 [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/medical-hands-holds-syringe-and-vaccine/199783080
- 5- Bianca. Family Financial Planning: Teaching Kids about Saving [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: [https://stock.adobe.com/nl/images/family-financial-planning-teaching-kids-about-saving/1236641272](https://stock.adobe.com/nl/images/family-financial-planning-teaching-kids-about-saving/1236641272)
- 6- Adobe Stock. A single yellow figure stands out amongst a crowd of blue figures [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/a-single-yellow-figure-stands-out-amongst-a-crowd-of-blue-figures/1147546461
7- Alina. Young kids with Down syndrome studying and playing in the room with white desks [Internet]. Adobe Stock; [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/young-kids-with-down-syndrome-studying-and-playing-in-the-room-with-white-desks/415837032
- 8- nyavileart P. Veterinarian or animal nurse is using a laptop to collect veterinary history and recommend a medical diagnosis in a hospital [Internet]. Adobe Stock; 2021 [cited 2025 Oct 5]. Available from: https://stock.adobe.com/nl/images/veterinarian-or-animal-nurse-is-using-a-laptop-to-collect-veterinary-history-and-recommend-a-medical-diagnosis-in-a-hospital-save-animals-care-and-treatment-animal-hospital/524507053
Blog Resources
- 1- CDC. Altered Immunocompetence [Internet]. Vaccines & Immunizations. 2024. Available from: Altered Immunocompetence
- 2- VACCINE SAFETY AND FALSE CONTRAINDICATIONS TO VACCINATION Training manual [Internet]. Available from: https://iris.who.int/bitstream/handle/10665/350968/WHO-EURO-2017-4683-44446-62833-eng.pdf?sequence=
- 3- Immunization and Other Considerations in Immunocompromised Children. 2024 Apr 25 [cited 2025 Aug 5];93–109. Available from: Immunization and Other Considerations in Immunocompromised Children
- 4- Esposito S, Cerutti M, Milani D, Menni F, Principi N. Vaccination coverage of children with rare genetic diseases and attitudes of their parents toward vaccines. Human Vaccines & Immunotherapeutics. 2015 Sep 4;12(3):801–5.
- 5- Sahn B, Lu Y, Hui‐Yuen JS, Fishbein J, Gottlieb BS, Eberhard BA, et al. The safety of COVID‐19 vaccination in immunocompromised children and young adults with immune‐mediated inflammatory disease. Acta pædiatrica. 2023 Jan 6;112(4):794–801.
- 6- Russo C, Otero A, Uranga M, Seery V, Raiden S, Algieri S, et al. Immunological memory to COVID-19 vaccines in immunocompromised and immunocompetent children. Frontiers in Cellular and Infection Microbiology. 2025 Feb 17;15.
- 7- Klein NP, Aukes L, Lee J, Fireman B, Shapira SK, Slade B, et al. Evaluation of Immunization Rates and Safety Among Children With Inborn Errors of Metabolism. Pediatrics [Internet]. 2011 May 1 [cited 2022 May 14];127(5):e1139–46. Available from: https://publications.aap.org/pediatrics/article-abstract/127/5/e1139/64912/Evaluation-of-Immunization-Rates-and-Safety-Among?redirectedFrom=fulltext
- 8- Zubarioglu T, Oral‐Cebeci S, Hopurcuoglu D, Uygur E, Ahmadzada S, Uzunyayla‐Inci G, et al. Safety of COVID ‐19 vaccines in children with inborn errors of metabolism in terms of developing metabolic decompensation. Journal of Paediatrics and Child Health. 2022 Sep 29;
- 9- The GLOBAL COVID-19 & Down Syndrome Vaccine Tracker | Global Down Syndrome Foundation [Internet]. Global Down Syndrome Foundation. 2024 [cited 2025 Aug 5]. Available from: The GLOBAL COVID-19 & Down Syndrome Vaccine Tracker | Global Down Syndrome Foundation
- 10- Centers for Disease Control and Prevention . People with Certain Medical Conditions Atlanta. Centers for Disease Control and Prevention; 2022.
- 11- Praveen N. K. Pitchan Velammal, Balasubramanian S, Fathima Shehnaz Ayoobkhan, Mohan K, Aggarwal P, Rabaan AA, et al. COVID‐19 in patients with Down syndrome: A systematic review. Immunity, inflammation and disease [Internet]. 2024 Mar 1 [cited 2024 Mar 26];12(3). Available from: NCBI - WWW Error Blocked Diagnostic
- 12- CDC. Altered Immunocompetence [Internet]. Vaccines & Immunizations. 2024. Available from: Altered Immunocompetence
- 13- Inborn errors of immunity, including primary immunodeficiency [Internet]. The Australian Immunisation Handbook. 2025. Available from: Inborn errors of immunity, including primary immunodeficiency
- 14- CDC. Child Immunization Schedule Notes [Internet]. Vaccines & Immunizations. 2024. Available from: Child Immunization Schedule Notes
- 15- Pittet LF, Posfay-Barbe KM. Vaccination of immune compromised children—an overview for physicians. European Journal of Pediatrics. 2021 Mar 5;
- 16- Morgan TM, Schlegel C, Edwards KM, Welch-Burke T, Zhu Y, Sparks R, et al. Vaccines Are Not Associated With Metabolic Events in Children With Urea Cycle Disorders. PEDIATRICS. 2011 Apr 11;127(5):e1147–53.
- 17- Bull MJ, Trotter T, Santoro SL, Christensen C, Grout RW. Health supervision for children and adolescents with down syndrome. Pediatrics [Internet]. 2022 Apr 18;149(5). Available from: Health Supervision for Children and Adolescents With Down Syndrome
- 18- Langkamp DL, Dusseau A, Brown MF. Vaccine Hesitancy and Low Immunization Rates in Children with Down Syndrome. The Journal of Pediatrics. 2020 Aug;223:64-67.e2.
- 19- Gravholt CH, Andersen NH, Christin-Maitre S, Davis SM, Duijnhouwer A, Gawlik A, Maciel-Guerra AT, Gutmark-Little I, Fleischer K, Hong D, Klein KO. Clinical practice guidelines for the care of girls and women with Turner syndrome: Proceedings from the 2023 Aarhus International Turner Syndrome Meeting. European Journal of Endocrinology. 2024 Jun;190(6):G53-151.
- 20- Pyle AK, George TN, Cummings JJ, Laventhal NT, Committee on Bioethics Tricot Laventhal Naomi MD, MA, FAAP Marie Geis Gina MD, FAAP Morgan Henderson Carrie MD, FAAP Susan Loeff Deborah MD, FAAP Michelson Kelly N. MD, MPH, FAAP Anne Ott Mary MD, MA, FAAP Taub Sara MD, FAAP. Guidance for Caring for Infants and Children With Trisomy 13 and Trisomy 18: Clinical Report. Pediatrics. 2025 Aug 1;156(2):e2025072719.
- 21- Vaccinations for people living with muscle wasting conditions - Muscular Dystrophy UK [Internet]. Muscular Dystrophy UK. 2025 [cited 2025 Aug 5]. Available from: Vaccinations for people living with muscle wasting conditions - Muscular Dystrophy UK
- 22- Australian Government Department of Health and Aged Care. Vaccination for People Who Are Immunocompromised [Internet]. The Australian Immunisation Handbook. 2022. Available from: Vaccination for people who are immunocompromised
- 23- Vaccination Recommendations [Internet]. Parent Project Muscular Dystrophy. Available from: Vaccination Recommendations
- 24- Wang LJ, Tsai CS, Chou WJ, Li CJ, Lee SY, Chen YC, et al. Medical outcomes of children with neurodevelopmental disorders after SARS-CoV-2 vaccination: A six-month follow-up study. Vaccine [Internet]. 2023 May 29;41(29):4267–73. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X2300628X
- 25- Epilepsy and immunisation - The Melbourne Vaccine Education Centre (MVEC) [Internet]. The Melbourne Vaccine Education Centre (MVEC). 2023. Available from: Epilepsy and immunisation - The Melbourne Vaccine Education Centre (MVEC)
- 26- Protocol for immunisation of children with Dravet syndrome or other vaccine-proximate seizures [Internet]. [cited 2025 Aug 5]. Available from: https://mvec.mcri.edu.au/wp-content/uploads/2021/03/Protocol-for-immunisation-of-children-with-Dravet-syndrome-or-other-vaccine-proximate-seizures_2.pdf
- 27- Australian Government Department of Health and Aged Care. Vaccination for People Who Are Immunocompromised [Internet]. The Australian Immunisation Handbook. 2022. Available from: Vaccination for people who are immunocompromised
- 28- Wexler M. Fragile X Patients Appear to Have Less Active Immune System, Study... [Internet]. Fragile X News Today. 2020 [cited 2025 Aug 5]. Available from: Fragile X Patients Appear to Have Less Active Immune System, Study…
- 29- Table. Recommended timing of live vaccine doses in adults and children taking corticosteroids [Internet]. The Australian Immunisation Handbook. 2024. Available from: Table. Recommended timing of live vaccine doses in adults and children taking corticosteroids
- 30- Gilbertson KE, Jackson HL, Dziuban EJ, Sherman SL, Berry-Kravis EM, Erickson CA, et al. Preventive care services and health behaviors in children with fragile X syndrome. Disability and Health Journal. 2019 Oct;12(4):564–73.
- 31- Medina T. Preventive Care Services in Fragile X Syndrome [Internet]. NFXF. 2019 [cited 2025 Aug 5]. Available from: Preventive Care Services in Fragile X Syndrome
- 32- COVID-19 Vaccines - The Ehlers Danlos Society [Internet]. The Ehlers Danlos Society. 2022 [cited 2025 Aug 5]. Available from: COVID-19 Vaccines - The Ehlers Danlos Society
- 33- Guerrerio AL, Mateja A, MacCarrick G, Fintzi J, Brittain E, Frischmeyer-Guerrerio PA, et al. Web-based survey investigating cardiovascular complications in hypermobile Ehlers-Danlos syndrome after COVID-19 infection and vaccination. PLoS ONE. 2024 Mar 21;19(3):e0298272–2.
- 34- Public Health Agency of Canada. Page 8: Canadian Immunization Guide: Part 1 - Key Immunization Information - [Internet]. Canada.ca. 2017. Available from: Vaccine administration practices: Canadian Immunization Guide
- 35- Marfan syndrome: MedlinePlus Genetics [Internet]. MedlinePlus - Health Information from the National Library of Medicine. Available from: Marfan syndrome: MedlinePlus Genetics
- 36- National institute of arthritis and musculoskeletal and skin diseases. Marfan syndrome [Internet]. National Institute of Arthritis and Musculoskeletal and Skin Diseases. 2019. Available from: Marfan Syndrome
- 37- New Diagnosis - Williams Syndrome Foundation [Internet]. Williams Syndrome Foundation. 2024 [cited 2025 Aug 5]. Available from: New Diagnosis - Williams Syndrome Foundation
- 38- Esposito S, Selicorni A, Daleno C, Valzano A, Cerutti M, Galeone C, et al. Immunogenicity, safety and tolerability of monovalent 2009 pandemic influenza A/H1N1 MF59-adjuvanted vaccine in children and adolescents with Williams or Cornelia De Lange syndrome. Human Vaccines. 2011 Jun;7(6):613–7.
- 39- Health Supervision for Children With Prader-Willi Syndrome. PEDIATRICS. 2010 Dec 27;127(1):195–204.
- 40- Clark K. Pneumonia Vaccinations and Prader-Willi Syndrome [Internet]. [cited 2025 Aug 5]. Available from: https://www.pwsausa.org/wp-content/uploads/2021/01/Pneumonia-vaccines_MA-108.pdf
- 41- COVID-19 Vaccination Recommendations for those with WS [Internet]. Williams Syndrome Association. 2021. Available from: COVID-19 Vaccination Recommendations for those with WS
- 42- Prader-Willi Syndrome Association (NZ) Incorporated PROVIDING ADVOCACY, EDUCATION AND SUPPORT FOR NEW ZEALANDERS WITH PWS AND THEIR FAMILIES [Internet]. PWS NEW ZELAND. 2022 [cited 2025 Aug 6]. Available from: https://www.pws.org.nz/wp-content/uploads/2022/03/PWSANZ-Antivirals-submission-final.pdf#:~:text=problems,disordered%20breathing
- 43- JHorsfall. Advice on COVID-19 vaccines for children [Internet]. Prader-Willi Syndrome Support and Information| PWSA UK . 2022 [cited 2025 Aug 6]. Available from: Advice on COVID-19 vaccines for children
- 44- Centers for Disease Control and Prevention. Child and adolescent immunization schedule by age [Internet]. Vaccines & Immunizations. 2025. Available from: Child and Adolescent Immunization Schedule by Age (Addendum updated August 7, 2025)
- 45- Cleveland Clinic. Childhood immunization schedule: Vaccines by age [Internet]. Cleveland Clinic. 2022. Available from: What Vaccines Does My Child Need, and at What Age?
- 46- Public Health Agency of Canada. Canadian Immunization Guide: Part 3 - Vaccination of Specific Populations - [Internet]. Canada.ca. 2016. Available from: Canadian Immunization Guide: Part 3. Vaccination of specific populations
- 47- Hofstetter AM, Jakob K, Klein NP, Dekker CL, Edwards KM, Halsey NA, et al. Live Vaccine Use and Safety in DiGeorge Syndrome. Pediatrics. 2014 Apr 1;133(4):e946–54.
- 48- Durusu Tanrıover M, Akar S, Turkcapar N, Karadag O, Ertenli I, Kiraz S. Vaccination recommendations for adult patients with rheumatic diseases. European Journal of Rheumatology. 2016 Feb 19;3(1):29–35.
- 49- CDC. How Vaccine Safety Monitoring Works [Internet]. Vaccine Safety Systems. 2024. Available from: How Vaccine Safety Monitoring Works
- 50- Howell KD, Kelly MM, DeMuri GP, McBride JA, Katz B, Edmonson MB, et al. COVID-19 Vaccination Intentions for Children With Medical Complexity. Hospital Pediatrics. 2022 Aug 30;12(9):e295–302.
- 51- WHO. Integrated people-centred care - GLOBAL [Internet]. www.who.int. World Health Organisation; 2023. Available from: Integrated people-centred care - GLOBAL
- 52- Ison MG. Vaccines for Use in Special Populations: Immunocompromised Hosts. The Journal of Infectious Diseases. 2024 Oct 15;
- 53- Xie J, Zi W, Li Z, He Y. Ontology-based Precision Vaccinology for Deep Mechanism Understanding and Precision Vaccine Development. Current Pharmaceutical Design [Internet]. 2021 Apr 2;27(7):900–10. Available from: Ontology-based Precision Vaccinology for Deep Mechanism Understanding and Precision Vaccine Development
- 54- World. Global childhood vaccination coverage holds steady, yet over 14 million infants remain unvaccinated – WHO, UNICEF [Internet]. Who.int. World Health Organization: WHO; 2025. Available from: Global childhood vaccination coverage holds steady, yet over 14 million infants remain unvaccinated – WHO, UNICEF
- 55- Barberi I, Venera Tiralongo, Marseglia L. Vaccination risks in children with rare diseases. The Italian Journal of Pediatrics/Italian journal of pediatrics [Internet]. 2015 Sep 30 [cited 2025 Aug 6];41(S2). Available from: Vaccination risks in children with rare diseases
- 56- Hofstetter AM, LaRussa P, Rosenthal SL. Vaccination of adolescents with chronic medical conditions: Special considerations and strategies for enhancing uptake. Human Vaccines & Immunotherapeutics. 2015 Jul 25;11(11):2571–81.
Enjoyed this article?
Share it with your friends on LinkedIn: Safe and Essential Vaccination for Children with Genetic Disorders
Follow us on LinkedIn for more updates and insights: Cima Care GmbH
